A systematic review of progranulin concentrations in biofluids in over 7,000 people-assessing the pathogenicity of GRN mutations and other influencing factors
- PMID: 38539243
- PMCID: PMC10976725
- DOI: 10.1186/s13195-024-01420-z
A systematic review of progranulin concentrations in biofluids in over 7,000 people-assessing the pathogenicity of GRN mutations and other influencing factors
Abstract
Background: Pathogenic heterozygous mutations in the progranulin gene (GRN) are a key cause of frontotemporal dementia (FTD), leading to significantly reduced biofluid concentrations of the progranulin protein (PGRN). This has led to a number of ongoing therapeutic trials aiming to treat this form of FTD by increasing PGRN levels in mutation carriers. However, we currently lack a complete understanding of factors that affect PGRN levels and potential variation in measurement methods. Here, we aimed to address this gap in knowledge by systematically reviewing published literature on biofluid PGRN concentrations.
Methods: Published data including biofluid PGRN concentration, age, sex, diagnosis and GRN mutation were collected for 7071 individuals from 75 publications. The majority of analyses (72%) had focused on plasma PGRN concentrations, with many of these (56%) measured with a single assay type (Adipogen) and so the influence of mutation type, age at onset, sex, and diagnosis were investigated in this subset of the data.
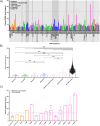
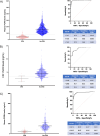
Results: We established a plasma PGRN concentration cut-off between pathogenic mutation carriers and non-carriers of 74.8 ng/mL using the Adipogen assay based on 3301 individuals, with a CSF concentration cut-off of 3.43 ng/mL. Plasma PGRN concentration varied by GRN mutation type as well as by clinical diagnosis in those without a GRN mutation. Plasma PGRN concentration was significantly higher in women than men in GRN mutation carriers (p = 0.007) with a trend in non-carriers (p = 0.062), and there was a significant but weak positive correlation with age in both GRN mutation carriers and non-carriers. No significant association was seen with weight or with TMEM106B rs1990622 genotype. However, higher plasma PGRN levels were seen in those with the GRN rs5848 CC genotype in both GRN mutation carriers and non-carriers.
Conclusions: These results further support the usefulness of PGRN concentration for the identification of the large majority of pathogenic mutations in the GRN gene. Furthermore, these results highlight the importance of considering additional factors, such as mutation type, sex and age when interpreting PGRN concentrations. This will be particularly important as we enter the era of trials for progranulin-associated FTD.
Keywords: Frontotemporal dementia; Progranulin.
© 2024. The Author(s).
Conflict of interest statement
HZ has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has given lectures in symposia sponsored by Alzecure, Biogen, Cellectricon, Fujirebio, Lilly, and Roche, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). JDR has served on medical advisory boards and consultancy for Alector, Arkuda Therapeutics, Wave Life Sciences, and Prevail Therapeutics. Consultancy for UCB, AC Immune, Astex Pharmaceuticals, Biogen, Takeda and Eisai. SKS has received author’s and advisory honoraria from Takeda/Shire and Medice Arzneimittel Pütter GmbH in the last 3 years. AL participated in advisory boards from Biogen, Eisai, Fujirebio-Europe, Grifols, Novartis, Roche, Otsuka Pharmaceutical, Nutricia, Zambón, y NovoNordisk. AL declares a filed patent application (WO2019175379 A1 Markers of synaptopathy in neurodegenerative disease). DA participated in advisory boards from Fujirebio-Europe, Roche Diagnostics, Grifols S.A. and Lilly, and received speaker honoraria from Fujirebio-Europe, Roche Diagnostics, Nutricia, Krka Farmacéutica S.L., Zambon S.A.U. and Esteve Pharmaceuticals S.A. DA declares a filed patent application (WO2019175379 A1 Markers of synaptopathy in neurodegenerative disease). MS has received consultancy honoraria from Ionis, UCB, Prevail, Orphazyme, Servier, Reata, GenOrph, AviadoBio, Biohaven, Zevra,and Lilly, all unrelated to the present manuscript. IIG participated in advisory boards from UCB Pharma S.A., and received speaker honoraria from Nutricia, Kern Pharma S., Krka Farmacéutica S.L., Zambon S.A.U. and Esteve Pharmaceuticals S.A.
Figures

References
-
- Arrant AE, Roth JR, Boyle NR, Kashyap SN, Hoffmann MQ, Murchison CF, Ramos EM, Nana AL, Spina S, Grinberg LT, Miller BL, Seeley WW, Roberson ED. Impaired β-glucocerebrosidase activity and processing in frontotemporal dementia due to progranulin mutations. Acta Neuropathol Commun. 2019;7(1):218. doi: 10.1186/s40478-019-0872-6. - DOI - PMC - PubMed
-
- Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M. Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature. 2006;442(7105):916–919. doi: 10.1038/nature05016. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 2019-02248/JPND GENFI-PROX grant
- 2019-02248/JPND GENFI-PROX grant
- 2019-02248/JPND GENFI-PROX grant
- 2019-02248/JPND GENFI-PROX grant
- 2019-02248/JPND GENFI-PROX grant
- 2019-02248/JPND GENFI-PROX grant
- MR/M008525/1/MRC Clinician Scientist Fellowship
- MR/M008525/1/MRC Clinician Scientist Fellowship
- 2013/017584/FAPESP grant number
- 01ED2008A/EU Joint Programme-Neurodegenerative Diseases networks Genfi-Prox
- 01ED2001/bPride
- FTLDc 01GI1007A, Moodmarker 01EW200/German Federal Ministry of Education and Research
- MIRIADE 860197, FAIR-PARK II 633190/the EU
- SFB1279/German Research Foundation/DFG
- D.3830/The foundation of the state Baden-Württemberg
- D.5009/Boehringer Ingelheim Ulm University BioCenter and the Thierry Latran Foundation
- #ALFGBG-71320/Swedish State Support for Clinical Research
- #201809-2016862/Alzheimer Drug Discovery Foundation (ADDF), USA
- #ADSF-21-831376-C, #ADSF-21-831381-C, #ADSF-21-831377-C/AD Strategic Fund and the Alzheimer's Association
- #2019-02397, #2022-01018/Swedish Research Council
- #FO2022-0270/the Bluefield Project, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden
- JPND2021-00694)/the European Union Joint Programme - Neurodegenerative Disease Research
- UKDRI-1003/UK Dementia Research Institute at UCL
- ARUK-RADF2021A-003/Alzheimer's Research UK
- BRC149/NS/MH/NIHR Rare Disease Translational Research Collaboration
- MR/M023664/1/MRC UK GENFI grant
- P30 AG062677/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous

