Liver-specific adiponectin gene therapy suppresses microglial NLRP3-inflammasome activation for treating Alzheimer's disease
- PMID: 38539253
- PMCID: PMC10967198
- DOI: 10.1186/s12974-024-03066-y
Liver-specific adiponectin gene therapy suppresses microglial NLRP3-inflammasome activation for treating Alzheimer's disease
Abstract
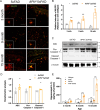
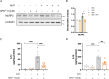
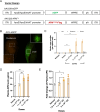
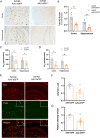
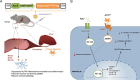
Adiponectin (APN) is an adipokine which predominantly expresses in adipocytes with neuroprotective and anti-inflammatory effects. We have recently indicated that circulatory trimeric APN can enter the brain by crossing the blood-brain barrier (BBB) and modulate microglia-mediated neuroinflammation. Here, we found that the microglial NLR family pyrin domain containing 3 (NLRP3)-inflammasome activation was exacerbated in APN-/-5xFAD mice in age-dependent manner. The focus of this study was to develop a new and tractable therapeutic approach for treating Alzheimer's disease (AD)-related pathology in 5xFAD mice using peripheral APN gene therapy. We have generated and transduced adeno-associated virus (AAV2/8) expressing the mouse mutated APN gene (APNC39S) into the liver of 5xFAD mice that generated only low-molecular-weight trimeric APN (APNTri). Single dose of AAV2/8-APNC39S in the liver increased circulatory and cerebral APN levels indicating the overexpressed APNTri was able to cross the BBB. Overexpression of APNTri decreased both the soluble and fibrillar Aβ in the brains of 5xFAD mice. AAV2/8-APNTri treatment reduced Aβ-induced IL-1β and IL-18 secretion by suppressing microglial NLRP3-inflammasome activation. The memory functions improved significantly in AAV-APNTri-treated 5xFAD mice with reduction of dystrophic neurites. These findings demonstrate that peripheral gene delivery to overexpress trimeric APN can be a potential therapy for AD.
Keywords: Adiponectin; Alzheimer’s Disease; Gene delivery; NLRP3; Neuroinflammation.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

