Targeting Oral Squamous Cell Carcinoma with Combined Polo-Like-Kinase-1 Inhibitors and γ-Radiation Therapy
- PMID: 38540116
- PMCID: PMC10968374
- DOI: 10.3390/biomedicines12030503
Targeting Oral Squamous Cell Carcinoma with Combined Polo-Like-Kinase-1 Inhibitors and γ-Radiation Therapy
Abstract
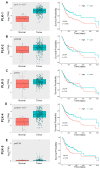
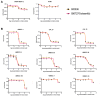
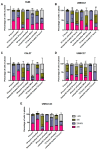
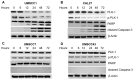
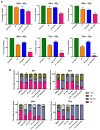
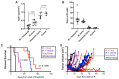
Polo-like-kinase-1 (PLK-1) is a serine/threonine kinase that regulates the cell cycle and acts as an oncogene in multiple cancers, including oral squamous cell carcinoma (OSCC). The loss of PLK-1 can inhibit growth and induce apoptosis, making it an attractive therapeutic target in OSCC. We evaluated the efficacy of PLK-1 inhibitors as novel, targeted therapeutics in OSCC. PLK-1 inhibition using BI6727 (volasertib) was found to affect cell death at low nanomolar concentrations in most tested OSCC cell lines, but not in normal oral keratinocytes. In cell lines resistant to volasertib alone, pre-treatment with radiotherapy followed by volasertib reduced cell viability and induced apoptosis. The combinatorial efficacy of volasertib and radiotherapy was replicated in xenograft mouse models. These findings highlight the potential of adding PLK-1 inhibitors to adjuvant therapy regimens in OSCC.
Keywords: OSCC; polo-like-kinase-1; volasertib; γ-radiation therapy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Bcl-2 degradation is an additional pro-apoptotic effect of polo-like kinase inhibition in cholangiocarcinoma cells.World J Gastroenterol. 2017 Jun 14;23(22):4007-4015. doi: 10.3748/wjg.v23.i22.4007. World J Gastroenterol. 2017. PMID: 28652654 Free PMC article.
-
Radiosensitization in esophageal squamous cell carcinoma: Effect of polo-like kinase 1 inhibition.Strahlenther Onkol. 2016 Apr;192(4):260-8. doi: 10.1007/s00066-016-0951-6. Epub 2016 Mar 7. Strahlenther Onkol. 2016. PMID: 26952039
-
Polo-like-kinase 1 (PLK-1) and c-myc inhibition with the dual kinase-bromodomain inhibitor volasertib in aggressive lymphomas.Oncotarget. 2017 Dec 6;8(70):114474-114480. doi: 10.18632/oncotarget.22967. eCollection 2017 Dec 29. Oncotarget. 2017. PMID: 29383095 Free PMC article.
-
Discovery and development of the Polo-like kinase inhibitor volasertib in cancer therapy.Leukemia. 2015 Jan;29(1):11-9. doi: 10.1038/leu.2014.222. Epub 2014 Jul 16. Leukemia. 2015. PMID: 25027517 Free PMC article. Review.
-
Spotlight on Volasertib: Preclinical and Clinical Evaluation of a Promising Plk1 Inhibitor.Med Res Rev. 2016 Jul;36(4):749-86. doi: 10.1002/med.21392. Epub 2016 May 3. Med Res Rev. 2016. PMID: 27140825 Review.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

