COVID-19 Clinical Features and Outcome in Italian Patients Treated with Biological Drugs Targeting Type 2 Inflammation
- PMID: 38541703
- PMCID: PMC10971220
- DOI: 10.3390/life14030378
COVID-19 Clinical Features and Outcome in Italian Patients Treated with Biological Drugs Targeting Type 2 Inflammation
Abstract
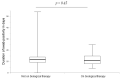
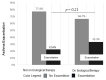
This is a multicentric investigation involving two Italian centers that examined the clinical course of COVID-19 in patients receiving biological therapy targeting type 2 inflammation and those not receiving biologicals. Since the beginning of the COVID-19 pandemic, the management of respiratory and allergic disorders and the potential impact of biological therapy in the most severe forms has been a point of uncertainty. Our multicentric investigation aimed to compare the clinical course of COVID-19 and the impact of vaccination in an Italian cohort of patients with atopic disorders caused by a type 2 inflammation, such as eosinophilic asthma, chronic rhinosinusitis with nasal polyposis (CRSwNP), atopic dermatitis (AD), and chronic spontaneous urticaria (CSU). A questionnaire was given to patients coming to our outpatient clinic for the first evaluation or follow-up visit, asking for the clinical characteristics of the infection, the ongoing therapy during the infection, any relevant change, and the patient's vaccination status. We enrolled 132 atopic patients from two Italian centers; 62 patients were on biological therapy at the time of infection (omalizumab 31%, mepolizumab 26%, benralizumab 19%, and dupilumab 24%). The median age was 56 (IQR 22.8) for patients on biologicals and 48 (IQR 26.5) for those not on biologicals (p = 0.028). The two groups were comparable in terms of sex, body mass index (BMI), smoking history, and systemic oral corticosteroid use (OCS). There were no significant differences in non-biological therapy and comorbidity between the two groups. The patients not on biological therapy had a prevalence of 87% for asthma, 52% for CRSwNP, 10% for CSU, and 6% for AD. The patients on biologicals had a prevalence of 93% for asthma, 17% for CRSwNP, and 10% for CSU. In our work, we observed that mAbs targeting type 2 inflammation in patients with COVID-19 appeared to be safe, with no worsening of symptoms, prolongation of infection, or increase in hospitalizations. Between the two groups, there were no significant differences in the duration of swab positivity (p = 0.45) and duration of symptoms (p = 0.38). During COVID-19, patients on biologicals experienced a significant increase in common cold-like symptoms (p = 0.038), dyspnea (p = 0.016), and more, but not significant, asthma exacerbations, with no significant differences between the different biologicals. Regarding the vaccination status, we observed that there was an increased number of hospitalizations among unvaccinated patients in both groups, although the difference did not reach statistical significance. No patients on biologicals reported safety issues or adverse effects associated with the use of biological treatments during COVID-19. Our investigation showed that mAbs against type 2 inflammation given during Coronavirus Disease 2019 are safe and do not impact the clinical course or main outcomes. Therefore, we found no signals suggesting that anti-Th2 biological therapy should be discontinued during SARS-CoV-2 infection. Controlled studies and analysis, including data from registries and real-life studies, are required to draw firm conclusions regarding the safety or possible advantages that anti-type 2 mAbs could offer in particular clinical contexts, such as infections.
Keywords: COVID-19; asthma; biologicals; type 2 inflammation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Use of biologicals in allergic and type-2 inflammatory diseases during the current COVID-19 pandemic: Position paper of Ärzteverband Deutscher Allergologen (AeDA)A, Deutsche Gesellschaft für Allergologie und Klinische Immunologie (DGAKI)B, Gesellschaft für Pädiatrische Allergologie und Umweltmedizin (GPA)C, Österreichische Gesellschaft für Allergologie und Immunologie (ÖGAI)D, Luxemburgische Gesellschaft für Allergologie und Immunologie (LGAI)E, Österreichische Gesellschaft für Pneumologie (ÖGP)F in co-operation with the German, Austrian, and Swiss ARIA groupsG, and the European Academy of Allergy and Clinical Immunology (EAACI)H.Allergol Select. 2020 Sep 7;4:53-68. doi: 10.5414/ALX02166E. eCollection 2020. Allergol Select. 2020. PMID: 32915172 Free PMC article.
-
COVID-19 vaccination of patients with allergies and type-2 inflammation with concurrent antibody therapy (biologicals) - A Position Paper of the German Society of Allergology and Clinical Immunology (DGAKI) and the German Society for Applied Allergology (AeDA).Allergol Select. 2021 Apr 1;5:140-147. doi: 10.5414/ALX02241E. eCollection 2021. Allergol Select. 2021. PMID: 33842829 Free PMC article.
-
Efficacy and safety of treatment with biologicals (benralizumab, dupilumab, mepolizumab, omalizumab and reslizumab) for severe eosinophilic asthma. A systematic review for the EAACI Guidelines - recommendations on the use of biologicals in severe asthma.Allergy. 2020 May;75(5):1023-1042. doi: 10.1111/all.14221. Epub 2020 Feb 24. Allergy. 2020. PMID: 32034960
-
State-of-the-art overview on biological treatment for CRSwNP.Rhinology. 2021 Apr 1;59(2):151-163. doi: 10.4193/Rhin20.570. Rhinology. 2021. PMID: 33459728 Review.
-
Biologicals in the Treatment of Pediatric Atopic Diseases.Handb Exp Pharmacol. 2020;261:131-151. doi: 10.1007/164_2019_307. Handb Exp Pharmacol. 2020. PMID: 32076895
Cited by
-
Cytokine Storm-Induced Thyroid Dysfunction in COVID-19: Insights into Pathogenesis and Therapeutic Approaches.Drug Des Devel Ther. 2024 Sep 20;18:4215-4240. doi: 10.2147/DDDT.S475005. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 39319193 Free PMC article. Review.
-
Efficacy of Dupilumab in the Treatment of Eosinophilic Esophagitis: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials.Life (Basel). 2025 Feb 17;15(2):307. doi: 10.3390/life15020307. Life (Basel). 2025. PMID: 40003715 Free PMC article. Review.
References
-
- Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271–280.e8. doi: 10.1016/j.cell.2020.02.052. - DOI - PMC - PubMed
-
- Sajuthi S.P., DeFord P., Li Y., Jackson N.D., Montgomery M.T., Everman J.L., Rios C.L., Pruesse E., Nolin J.D., Plender E.G., et al. Type 2 and interferon inflammation regulate SARS-CoV-2 entry factor expression in the airway epithelium. Nat. Commun. 2020;11:5139. doi: 10.1038/s41467-020-18781-2. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous