Membrane Interaction Characteristics of the RTX Toxins and the Cholesterol-Dependence of Their Cytolytic/Cytotoxic Activity
- PMID: 38542105
- PMCID: PMC10970614
- DOI: 10.3390/ijms25063131
Membrane Interaction Characteristics of the RTX Toxins and the Cholesterol-Dependence of Their Cytolytic/Cytotoxic Activity
Abstract
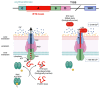
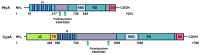
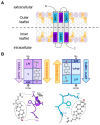
RTX toxins are important virulence factors produced by a wide range of Gram-negative bacteria. They are secreted as water-soluble proteins that are able to bind to the host cell membrane and insert hydrophobic segments into the lipid bilayer that ultimately contribute to the formation of transmembrane pores. Ion diffusion through these pores leads then to cytotoxic and cytolytic effects on the hosts. Several reports have evidenced that the binding of several RTX toxins to the target cell membrane may take place through a high-affinity interaction with integrins of the β2 family that is highly expressed in immune cells of the myeloid lineage. However, at higher toxin doses, cytotoxicity by most RTX toxins has been observed also on β2-deficient cells in which toxin binding to the cell membrane has been proposed to occur through interaction with glycans of glycosylated lipids or proteins present in the membrane. More recently, cumulative pieces of evidence show that membrane cholesterol is essential for the mechanism of action of several RTX toxins. Here, we summarize the most important aspects of the RTX toxin interaction with the target cell membrane, including the cholesterol dependence, the recent identification in the sequences of several RTX toxins of linear motifs coined as the Cholesterol Recognition/interaction Amino acid Consensus (CRAC), and the reverse or mirror CARC motif, which is involved in the toxin-cholesterol interaction.
Keywords: RTX toxins; cholesterol; cholesterol-binding motifs; lipid-protein interactions; pore-forming toxins.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity.Biochim Biophys Acta. 2016 Mar;1858(3):526-37. doi: 10.1016/j.bbamem.2015.10.025. Epub 2015 Oct 30. Biochim Biophys Acta. 2016. PMID: 26523409 Review.
-
Aggregatibacter actinomycetemcomitans leukotoxin utilizes a cholesterol recognition/amino acid consensus site for membrane association.J Biol Chem. 2013 Aug 9;288(32):23607-21. doi: 10.1074/jbc.M113.486654. Epub 2013 Jun 21. J Biol Chem. 2013. PMID: 23792963 Free PMC article.
-
Membrane Permeabilization by Pore-Forming RTX Toxins: What Kind of Lesions Do These Toxins Form?Toxins (Basel). 2019 Jun 18;11(6):354. doi: 10.3390/toxins11060354. Toxins (Basel). 2019. PMID: 31216745 Free PMC article. Review.
-
Cholesterol stimulates the lytic activity of Adenylate Cyclase Toxin on lipid membranes by promoting toxin oligomerization and formation of pores with a greater effective size.FEBS J. 2021 Dec;288(23):6795-6814. doi: 10.1111/febs.16107. Epub 2021 Jul 14. FEBS J. 2021. PMID: 34216517 Free PMC article.
-
Pore forming activity of the potent RTX-toxin produced by pediatric pathogen Kingella kingae: Characterization and comparison to other RTX-family members.Biochim Biophys Acta. 2015 Jul;1848(7):1536-44. doi: 10.1016/j.bbamem.2015.03.036. Epub 2015 Apr 7. Biochim Biophys Acta. 2015. PMID: 25858109 Free PMC article.
References
-
- Linhartová I., Bumba L., Mašín J., Basler M., Osička R., Kamanová J., Procházková K., Adkins I., Hejnová-Holubová J., Sadílková L., et al. RTX Proteins: A Highly Diverse Family Secreted by a Common Mechanism. FEMS Microbiol. Rev. 2010;34:1076–1112. doi: 10.1111/j.1574-6976.2010.00231.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

