A Review of Approaches to Potentiate the Activity of Temozolomide against Glioblastoma to Overcome Resistance
- PMID: 38542190
- PMCID: PMC10970334
- DOI: 10.3390/ijms25063217
A Review of Approaches to Potentiate the Activity of Temozolomide against Glioblastoma to Overcome Resistance
Abstract
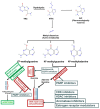
A glioblastoma (GBM) is one of the most aggressive, infiltrative, and treatment-resistant malignancies of the central nervous system (CNS). The current standard of care for GBMs include maximally safe tumor resection, followed by concurrent adjuvant radiation treatment and chemotherapy with the DNA alkylating agent temozolomide (TMZ), which was approved by the FDA in 2005 based on a marginal increase (~2 months) in overall survival (OS) levels. This treatment approach, while initially successful in containing and treating GBM, almost invariably fails to prevent tumor recurrence. In addition to the limited therapeutic benefit, TMZ also causes debilitating adverse events (AEs) that significantly impact the quality of life of GBM patients. Some of the most common AEs include hematologic (e.g., thrombocytopenia, neutropenia, anemia) and non-hematologic (e.g., nausea, vomiting, constipation, dizziness) toxicities. Recurrent GBMs are often resistant to TMZ and other DNA-damaging agents. Thus, there is an urgent need to devise strategies to potentiate TMZ activity, to overcome drug resistance, and to reduce dose-dependent AEs. Here, we analyze major mechanisms of the TMZ resistance-mediated intracellular signaling activation of DNA repair pathways and the overexpression of drug transporters. We review some of the approaches investigated to counteract these mechanisms of resistance to TMZ, including the use of chemosensitizers and drug delivery strategies to enhance tumoral drug exposure.
Keywords: combination therapeutics; glioblastoma; treatment resistance.
Conflict of interest statement
Author Nimita Dave was employed by the company Lapix Therapeutics. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Louis D.N., Perry A., Wesseling P., Brat D.J., Cree I.A., Figarella-Branger D., Hawkins C., Ng H.K., Pfister S.M., Reifenberger G., et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro Oncol. 2021;23:1231–1251. doi: 10.1093/neuonc/noab106. - DOI - PMC - PubMed
-
- Verhaak R.G.W., Hoadley K.A., Purdom E., Wang V., Wilkerson M.D., Miller C.R., Ding L., Golub T., Jill P., Alexe G., et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. 2010;17:98–110. doi: 10.1016/j.ccr.2009.12.020. - DOI - PMC - PubMed
-
- Wang Q., Hu B., Hu X., Kim H., Squatrito M., Scarpace L., deCarvalho A.C., Lyu S., Li P., Li Y., et al. Tumor Evolution of Glioma-Intrinsic Gene Expression Subtypes Associates with Immunological Changes in the Microenvironment. Cancer Cell. 2018;32:42–56.e6. doi: 10.1016/j.ccell.2017.06.003. Erratum in Cancer Cell 2018, 33, 152. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

