Beyond Quiescent and Active: Intermediate Microglial Transcriptomic States in a Mouse Model of Down Syndrome
- PMID: 38542263
- PMCID: PMC10970051
- DOI: 10.3390/ijms25063289
Beyond Quiescent and Active: Intermediate Microglial Transcriptomic States in a Mouse Model of Down Syndrome
Abstract
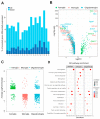
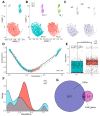
Research on microglia in Down syndrome (DS) has shown that microglial activation, increased inflammatory gene expression, and oxidative stress occur at different ages in DS brains. However, most studies resulted in simplistic definitions of microglia as quiescent or active, ignoring potential intermediate states. Indeed, recent work on microglial cells in young DS brains indicated that those evolve through different intermediate activation phenotypes before reaching a fully activated state. Here we used single nucleus RNA sequencing, to examine how trisomy affects microglial states in the Ts65Dn mouse model of DS. Despite no substantial changes in the proportion of glial populations, differential expression analysis revealed cell type-specific gene expression changes, most notably in astroglia, microglia, and oligodendroglia. Focusing on microglia, we identified differential expression of genes associated with different microglial states, including disease-associated microglia (DAMs), activated response microglia (ARMs), and human Alzheimer's disease microglia (HAMs), in trisomic microglia. Furthermore, pseudotime analysis reveals a unique reactivity profile in Ts65Dn microglia, with fewer in a homeostatic state and more in an intermediate aberrantly reactive state than in euploid microglia. This comprehensive understanding of microglial transcriptional dynamics sheds light on potential pathogenetic mechanisms but also possible avenues for therapy for neurodevelopmental disorders.
Keywords: Down syndrome; disease-associated microglia; microglia.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Altered cell and RNA isoform diversity in aging Down syndrome brains.Proc Natl Acad Sci U S A. 2021 Nov 23;118(47):e2114326118. doi: 10.1073/pnas.2114326118. Proc Natl Acad Sci U S A. 2021. PMID: 34795060 Free PMC article.
-
Quantitative proteomics of acutely-isolated mouse microglia identifies novel immune Alzheimer's disease-related proteins.Mol Neurodegener. 2018 Jun 28;13(1):34. doi: 10.1186/s13024-018-0266-4. Mol Neurodegener. 2018. PMID: 29954413 Free PMC article.
-
Type-I-interferon signaling drives microglial dysfunction and senescence in human iPSC models of Down syndrome and Alzheimer's disease.Cell Stem Cell. 2022 Jul 7;29(7):1135-1153.e8. doi: 10.1016/j.stem.2022.06.007. Cell Stem Cell. 2022. PMID: 35803230 Free PMC article.
-
The interplay between microglial states and major risk factors in Alzheimer's disease through the eyes of single-cell RNA-sequencing: beyond black and white.J Neurophysiol. 2019 Oct 1;122(4):1291-1296. doi: 10.1152/jn.00395.2019. Epub 2019 Jul 31. J Neurophysiol. 2019. PMID: 31365320 Review.
-
Microglia heterogeneity and neurodegeneration: The emerging paradigm of the role of immunity in Alzheimer's disease.J Neuroimmunol. 2020 Apr 15;341:577185. doi: 10.1016/j.jneuroim.2020.577185. Epub 2020 Feb 3. J Neuroimmunol. 2020. PMID: 32045774 Review.
Cited by
-
Mass-Guided Single-Cell MALDI Imaging of Low-Mass Metabolites Reveals Cellular Activation Markers.Adv Sci (Weinh). 2025 Feb;12(5):e2410506. doi: 10.1002/advs.202410506. Epub 2024 Dec 12. Adv Sci (Weinh). 2025. PMID: 39665230 Free PMC article.
References
-
- Guidi S., Bonasoni P., Ceccarelli C., Santini D., Gualtieri F., Ciani E., Bartesaghi R. Neurogenesis impairment and increased cell death reduce total neuron number in the hippocampal region of fetuses with Down syndrome. Brain Pathol. 2008;18:180–197. doi: 10.1111/j.1750-3639.2007.00113.x. - DOI - PMC - PubMed
-
- Pinto B., Morelli G., Rastogi M., Savardi A., Fumagalli A., Petretto A., Petretto A., Bartolucci M., Varea E., Catelani T., et al. Rescuing Over-activated Microglia Restores Cognitive Performance in Juvenile Animals of the Dp(16) Mouse Model of Down Syndrome. Neuron. 2020;108:887–904.e12. doi: 10.1016/j.neuron.2020.09.010. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

