Long Non-Coding RNA-Cardiac-Inducing RNA 6 Mediates Repair of Infarcted Hearts by Inducing Mesenchymal Stem Cell Differentiation into Cardiogenic Cells through Cyclin-Dependent Kinase 1
- PMID: 38542439
- PMCID: PMC10971150
- DOI: 10.3390/ijms25063466
Long Non-Coding RNA-Cardiac-Inducing RNA 6 Mediates Repair of Infarcted Hearts by Inducing Mesenchymal Stem Cell Differentiation into Cardiogenic Cells through Cyclin-Dependent Kinase 1
Abstract
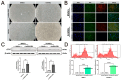
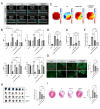
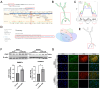
This study aims to investigate the induction effect of LncRNA-CIR6 on MSC differentiation into cardiogenic cells in vitro and in vivo. In addition to pretreatment with Ro-3306 (a CDK1 inhibitor), LncRNA-CIR6 was transfected into BMSCs and hUCMSCs using jetPRIME. LncRNA-CIR6 was further transfected into the hearts of C57BL/6 mice via 100 μL of AAV9-cTnT-LncRNA-CIR6-ZsGreen intravenous injection. After three weeks of transfection followed by AMI surgery, hUCMSCs (5 × 105/100 μL) were injected intravenously one week later. Cardiac function was evaluated using VEVO 2100 and electric mapping nine days after cell injection. Immunofluorescence, Evans blue-TTC, Masson staining, FACS, and Western blotting were employed to determine relevant indicators. LncRNA-CIR6 induced a significant percentage of differentiation in BMSCs (83.00 ± 0.58)% and hUCMSCs (95.43 ± 2.13)% into cardiogenic cells, as determined by the expression of cTnT using immunofluorescence and FACS. High cTNT expression was observed in MSCs after transfection with LncRNA-CIR6 by Western blotting. Compared with the MI group, cardiac contraction and conduction function in MI hearts treated with LncRNA-CIR6 or combined with MSCs injection groups were significantly increased, and the areas of MI and fibrosis were significantly lower. The transcriptional expression region of LncRNA-CIR6 was on Chr17 from 80209290 to 80209536. The functional region of LncRNA-CIR6 was located at nucleotides 0-50/190-255 in the sequence. CDK1, a protein found to be related to the proliferation and differentiation of cardiomyocytes, was located in the functional region of the LncRNA-CIR6 secondary structure (from 0 to 17). Ro-3306 impeded the differentiation of MSCs into cardiogenic cells, while MSCs transfected with LncRNA-CIR6 showed a high expression of CDK1. LncRNA-CIR6 mediates the repair of infarcted hearts by inducing MSC differentiation into cardiogenic cells through CDK1.
Keywords: BMSCs; CDK1; LncRNA-CIR6; hUMSCs; myocardial infarction.
Conflict of interest statement
All authors declare that they have no conflicts of interest.
Figures




Similar articles
-
Innovative Therapeutic Strategies for Myocardial Infarction Across Various Stages: Non-Coding RNA and Stem Cells.Int J Mol Sci. 2024 Dec 30;26(1):231. doi: 10.3390/ijms26010231. Int J Mol Sci. 2024. PMID: 39796085 Free PMC article. Review.
-
Atorvastatin enhances the therapeutic efficacy of mesenchymal stem cells-derived exosomes in acute myocardial infarction via up-regulating long non-coding RNA H19.Cardiovasc Res. 2020 Feb 1;116(2):353-367. doi: 10.1093/cvr/cvz139. Cardiovasc Res. 2020. PMID: 31119268 Free PMC article.
-
Long noncoding RNA Braveheart promotes cardiogenic differentiation of mesenchymal stem cells in vitro.Stem Cell Res Ther. 2017 Jan 17;8(1):4. doi: 10.1186/s13287-016-0454-5. Stem Cell Res Ther. 2017. PMID: 28095922 Free PMC article.
-
Bone-derived Nestin-positive mesenchymal stem cells improve cardiac function via recruiting cardiac endothelial cells after myocardial infarction.Stem Cell Res Ther. 2019 Apr 27;10(1):127. doi: 10.1186/s13287-019-1217-x. Stem Cell Res Ther. 2019. PMID: 31029167 Free PMC article.
-
AKT-modified autologous intracoronary mesenchymal stem cells prevent remodeling and repair in swine infarcted myocardium.Chin Med J (Engl). 2010 Jul;123(13):1702-8. Chin Med J (Engl). 2010. PMID: 20819633
Cited by
-
Engineering mesenchymal stem cells for premature ovarian failure: overcoming challenges and innovating therapeutic strategies.Theranostics. 2024 Oct 7;14(17):6487-6515. doi: 10.7150/thno.102641. eCollection 2024. Theranostics. 2024. PMID: 39479455 Free PMC article. Review.
-
ELABELA promotes the migration and homing of bone marrow mesenchymal stem cells to myocardial injury sites through the ERK1/2/miR-299a-5p/Exo70 pathway.Front Pharmacol. 2025 Feb 3;16:1541869. doi: 10.3389/fphar.2025.1541869. eCollection 2025. Front Pharmacol. 2025. PMID: 39963243 Free PMC article.
-
Innovative Therapeutic Strategies for Myocardial Infarction Across Various Stages: Non-Coding RNA and Stem Cells.Int J Mol Sci. 2024 Dec 30;26(1):231. doi: 10.3390/ijms26010231. Int J Mol Sci. 2024. PMID: 39796085 Free PMC article. Review.
-
Developmental and Evolutionary Heart Adaptations Through Structure-Function Relationships.J Cardiovasc Dev Dis. 2025 Feb 22;12(3):83. doi: 10.3390/jcdd12030083. J Cardiovasc Dev Dis. 2025. PMID: 40137081 Free PMC article. Review.
References
-
- Penha E.M., Aguiar P.H., Barrouin-Melo S.M., de Lima R.S., da Silveira A.C., Otelo A.R., Pinheiro C.M., Ribeiro-Dos-Santos R., Soares M.B. Clinical neurofunctional rehabilitation of a cat with spinal cord injury after hemilaminectomy and autologous stem cell transplantation. Int. J. Stem Cells. 2012;5:146–150. doi: 10.15283/ijsc.2012.5.2.146. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

