Suppression of Borna Disease Virus Replication during Its Persistent Infection Using the CRISPR/Cas13b System
- PMID: 38542493
- PMCID: PMC10971351
- DOI: 10.3390/ijms25063523
Suppression of Borna Disease Virus Replication during Its Persistent Infection Using the CRISPR/Cas13b System
Abstract
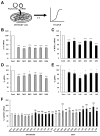
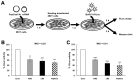
Borna disease virus (BoDV-1) is a bornavirus that infects the central nervous systems of various animal species, including humans, and causes fatal encephalitis. BoDV-1 also establishes persistent infection in neuronal cells and causes neurobehavioral abnormalities. Once neuronal cells or normal neural networks are lost by BoDV-1 infection, it is difficult to regenerate damaged neural networks. Therefore, the development of efficient anti-BoDV-1 treatments is important to improve the outcomes of the infection. Recently, one of the clustered regularly interspaced short palindromic repeats (CRISPRs) and CRISPR-associated (Cas) systems, CRISPR/Cas13, has been utilized as antiviral tools. However, it is still unrevealed whether the CRISPR/Cas13 system can suppress RNA viruses in persistently infected cells. In this study, we addressed this question using persistently BoDV-1-infected cells. The CRISPR/Cas13 system targeting viral mRNAs efficiently decreased the levels of target viral mRNAs and genomic RNA (gRNA) in persistently infected cells. Furthermore, the CRISPR/Cas13 system targeting viral mRNAs also suppressed BoDV-1 infection if the system was introduced prior to the infection. Collectively, we demonstrated that the CRISPR/Cas13 system can suppress BoDV-1 in both acute and persistent infections. Our findings will open the avenue to treat prolonged infection with RNA viruses using the CRISPR/Cas13 system.
Keywords: Borna disease virus; CRISPR/Cas13b; antiviral; antivirals; persistent infection.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
The regulation of persistent Borna disease virus infection by RNA silencing factors in human cells.Biochem Biophys Res Commun. 2023 May 28;658:122-127. doi: 10.1016/j.bbrc.2023.03.069. Epub 2023 Mar 31. Biochem Biophys Res Commun. 2023. PMID: 37030066
-
Generation of a non-transmissive Borna disease virus vector lacking both matrix and glycoprotein genes.Microbiol Immunol. 2017 Sep;61(9):380-386. doi: 10.1111/1348-0421.12505. Microbiol Immunol. 2017. PMID: 28776750
-
ADAR2 Is Involved in Self and Nonself Recognition of Borna Disease Virus Genomic RNA in the Nucleus.J Virol. 2020 Feb 28;94(6):e01513-19. doi: 10.1128/JVI.01513-19. Print 2020 Feb 28. J Virol. 2020. PMID: 31852792 Free PMC article.
-
Reverse Genetics and Artificial Replication Systems of Borna Disease Virus 1.Viruses. 2022 Oct 12;14(10):2236. doi: 10.3390/v14102236. Viruses. 2022. PMID: 36298790 Free PMC article. Review.
-
Molecular Mechanisms of RNA Targeting by Cas13-containing Type VI CRISPR-Cas Systems.J Mol Biol. 2019 Jan 4;431(1):66-87. doi: 10.1016/j.jmb.2018.06.029. Epub 2018 Jun 22. J Mol Biol. 2019. PMID: 29940185 Review.
Cited by
-
mRNA-encoded Cas13 treatment of Influenza via site-specific degradation of genomic RNA.PLoS Pathog. 2024 Jul 5;20(7):e1012345. doi: 10.1371/journal.ppat.1012345. eCollection 2024 Jul. PLoS Pathog. 2024. PMID: 38968329 Free PMC article.
References
-
- Honda T. Bornavirus infection in human diseases and its molecular neuropathology. Clin. Exp. Neuroimmunol. 2022;13:7–16. doi: 10.1111/cen3.12686. - DOI
-
- Schulze V., Große R., Fürstenau J., Forth L.F., Ebinger A., Richter M.T., Tappe D., Mertsch T., Klose K., Schlottau K., et al. Borna disease outbreak with high mortality in an alpaca herd in a previously unreported endemic area in Germany. Transbound. Emerg. Dis. 2020;67:2093–2107. doi: 10.1111/tbed.13556. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

