Stereoselective [4+3]-Cycloaddition of 2-Amino-β-nitrostyrenes with Azaoxyallyl Cations to Access Functionalized 1,4-Benzodiazepin-3-ones
- PMID: 38542858
- PMCID: PMC10975549
- DOI: 10.3390/molecules29061221
Stereoselective [4+3]-Cycloaddition of 2-Amino-β-nitrostyrenes with Azaoxyallyl Cations to Access Functionalized 1,4-Benzodiazepin-3-ones
Abstract
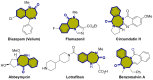
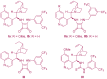
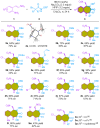
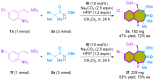
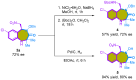
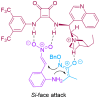
The 1,4-benzodiazepine structural framework is a fascinating element commonly found in biologically active and pharmaceutically relevant compounds. A highly efficient method for synthesizing 1,4-benzodiazepin-3-ones is described, involving a [4+3]-cycloaddition reaction between 2-amino-β-nitrostyrenes and α-bromohydroxamate, with Cs2CO3 used as a base. This process yielded the desired 1,4-benzodiazepines in good yields. Furthermore, an organocatalytic asymmetric [4+3]-cycloaddition was successfully accomplished using a bifunctional squaramide-based catalyst. This approach enabled the enantioselective synthesis of chiral 1,4-benzodiazepines with commendable yields and moderate enantioselectivities, reaching up to 80% yield and 72% ee.
Keywords: 1,4-benzodiazepine; [4+3]-cycloaddition; asymmetric catalysis; azaoxyallyl cation; organocatalysis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
[4+2]-Cycloaddition of 2-Aminophenyl Enones with Cyclic N-Sulfimines to Access Enantioenriched Ring-Fused Aminals.Chem Asian J. 2024 Feb 1;19(3):e202301011. doi: 10.1002/asia.202301011. Epub 2023 Nov 30. Chem Asian J. 2024. PMID: 37984888
-
Organocatalytic Enantioselective Synthesis of Polycyclic Benzosultams from 2-Amino-β-nitrostyrenes with Cyclic N-Sulfonyl Ketimines.Org Lett. 2024 Feb 23;26(7):1472-1477. doi: 10.1021/acs.orglett.4c00137. Epub 2024 Feb 13. Org Lett. 2024. PMID: 38349156
-
Construction of Axially Chiral Compounds via Asymmetric Organocatalysis.Acc Chem Res. 2018 Feb 20;51(2):534-547. doi: 10.1021/acs.accounts.7b00602. Epub 2018 Feb 8. Acc Chem Res. 2018. PMID: 29419282
-
Asymmetric Organocatalytic [3 + 2]-Cycloaddition of N-Alkoxy-4-oxo-acrylamides with Isatin-Derived Ketimines: Access to Enantioselective Synthesis of Spirooxindole-imidazolidinones.Org Lett. 2025 May 9;27(18):4706-4711. doi: 10.1021/acs.orglett.5c01076. Epub 2025 Apr 27. Org Lett. 2025. PMID: 40289503
-
Organocatalytic asymmetric Michael addition of α-alkylidene succinimides to nitrostyrenes.Org Biomol Chem. 2016 Jul 14;14(26):6337-45. doi: 10.1039/c6ob00711b. Epub 2016 Jun 8. Org Biomol Chem. 2016. PMID: 27272983
References
-
- Magi E., Fattorusso C., Persico M., Corvino A., Esposito G., Fiorino F., Luciano P., Perissutti E., Santagada V., Severino B., et al. New Insights into the Structure−Activity Relationship and Neuroprotective Profile of Benzodiazepinone Derivatives of Neurounina-1 as Modulators of the Na+/Ca2+ Exchanger Isoforms. J. Med. Chem. 2021;64:17901–17919. doi: 10.1021/acs.jmedchem.1c01212. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

