Selective Generation of Aldimine and Ketimine Tautomers of the Schiff Base Condensates of Amino Acids with Imidazole Aldehydes or of Imidazole Methanamines with Pyruvates-Isomeric Control with 2- vs. 4-Substituted Imidazoles
- PMID: 38542960
- PMCID: PMC10974184
- DOI: 10.3390/molecules29061324
Selective Generation of Aldimine and Ketimine Tautomers of the Schiff Base Condensates of Amino Acids with Imidazole Aldehydes or of Imidazole Methanamines with Pyruvates-Isomeric Control with 2- vs. 4-Substituted Imidazoles
Abstract
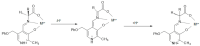
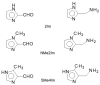
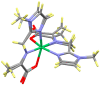
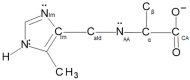
The Schiff base condensation of 5-methyl-4-imidazole carboxaldehyde, 5Me4ImCHO, and the anion of an amino acid, H2N-CH(R)CO2- (R = -CH3, -CH(CH3)2 and -CH2CH(CH3)2), gives the aldimine tautomer, Im-CH=N-CH(R)CO2-, while that of 5-methylimidazole-4-methanamine, 5MeIm-4-CH2NH2, with a 2-oxocarboxylate anion, R-C(O)-CO2-, gives the isomeric ketimine tautomer, Im-CH2-N=C(R)CO2-. All are isolated as the neutral nickel(II) complexes, NiL2, and are characterized by single crystal structure determination, IR, and positive ion ESI MS. In the cases of the 4 substituted imidazoles, either 5MeIm-4-CHO or 5MeIm-4-CH2NH2, both the aldimine and ketimine complexes are isolated cleanly with no evidence of an equilibrium between the two tautomers under the experimental conditions. The aldimines are blue while the tautomeric ketimines are green. In contrast, for the 2-substituted imidazoles, with either Im-2-CHO or Im-2-CH2NH2, the isolated product from the Schiff base condensation is the ketimine, which in the solid is green, as observed for the 4-isomer. These results suggest that for the 2-substituted imidazoles, there is a facile equilibrium between the aldimine and ketimine tautomers, and that the ketimine form is the thermodynamically favored tautomer. The aldimine tautomers of the 4-substituted imidazoles have three stereogenic centers, the nickel (Δ or Ʌ) and the two alpha carbon atoms (R or S). The observed pair of enantiomers is the ɅRR/ΔSS enantiomeric pair, suggesting that this pair is lower in energy than the others and that this is in general the preferred chiral correlation in these complexes.
Keywords: Schiff base; aldimine; amino acids; homochirality; imidazole; ketimine; pyruvates; tautomerization.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
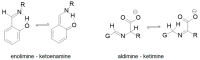







Similar articles
-
Time-resolved fluorescence of O-acetylserine sulfhydrylase catalytic intermediates.Biochemistry. 1997 Dec 9;36(49):15419-27. doi: 10.1021/bi970464i. Biochemistry. 1997. PMID: 9398272
-
1H-NMR study on the tautomerism of the imidazole ring of histidine residues. I. Microscopic pK values and molar ratios of tautomers in histidine-containing peptides.Biochim Biophys Acta. 1983 Feb 15;742(3):576-85. doi: 10.1016/0167-4838(83)90276-5. Biochim Biophys Acta. 1983. PMID: 6838890
-
A change in the internal aldimine lysine (K42) in O-acetylserine sulfhydrylase to alanine indicates its importance in transimination and as a general base catalyst.Biochemistry. 1996 Oct 15;35(41):13485-93. doi: 10.1021/bi961517j. Biochemistry. 1996. PMID: 8873618
-
Reversible-irreversible approach to Schiff base macrocycles: access to isomeric macrocycles with multiple salphen pockets.Org Lett. 2008 Mar 20;10(6):1255-8. doi: 10.1021/ol8001317. Epub 2008 Feb 21. Org Lett. 2008. PMID: 18288856
-
The low energy tautomers and conformers of the dipeptides HisGly and GlyHis and of their sodium ion complexes in the gas phase.Phys Chem Chem Phys. 2005 Nov 7;7(21):3744-55. doi: 10.1039/b508092d. Epub 2005 Sep 9. Phys Chem Chem Phys. 2005. PMID: 16358024
Cited by
-
Chirality Transfer and Oxazolidine Formation in Reaction of L and D Enantiomers of β-Hydroxy Amino Acids with Nitrogenous Carboxaldehydes and Nickel(II).Molecules. 2025 Jul 10;30(14):2913. doi: 10.3390/molecules30142913. Molecules. 2025. PMID: 40733184 Free PMC article.
References
-
- Ikuta Y., Ooidemizu M., Yamahata Y., Yamada M., Osa S., Matsumoto N., Iijima S., Sunatsuki Y., Kojima M., Dahan F., et al. A New Family of Spin Crossover Complexes with a Tripod Ligand Containing Three Imidazoles: Synthesis, Characterization, and Magnetic Properties of [FeIIH3LMe](NO3)2·1.5H2O, [FeIIILMe]·3.5H2O, [FeIIH3LMe][FeIILMe]NO3, and [FeIIH3LMe][FeIIILMe](NO3)2 (H3LMe = Tris [2-(((2-methylimidazol-4-yll)methylidene)amino)ethyl]amine) Inorg. Chem. 2003;42:7001–7017. - PubMed
-
- Yamada M., Ooidemizu M., Ikuta Y., Osa S., Matsumoto N., Iijima S., Kojima M., Dahan F., Tuchagues J.P. Interlayer Interaction of Two-Dimensional Layered Spin Crossover Complexes [FeIIH3LMe][FeIILMe]X (X− = ClO4−, BF4−, PF6−, AsF6−, and SbF6−; H3LMe = Tris [2-(((2-methylimidazol-4-yl)methylidene)amino)ethyl]amine) Inorg. Chem. 2003;42:8406–8416. doi: 10.1021/ic034439e. - DOI - PubMed
-
- Brewer G. Structural Evidence of Spin State Selection and Spin Crossover Behavior of Tripodal Schiff Base Complexes of tris(2-aminoethyl)amine and Related Tripodal Amines. Magnetochemistry. 2020;6:28. doi: 10.3390/magnetochemistry6020028. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

