Evaluation of Pyrophosphate-Driven Proton Pumps in Saccharomyces cerevisiae under Stress Conditions
- PMID: 38543676
- PMCID: PMC10976212
- DOI: 10.3390/microorganisms12030625
Evaluation of Pyrophosphate-Driven Proton Pumps in Saccharomyces cerevisiae under Stress Conditions
Abstract
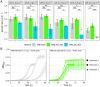
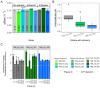
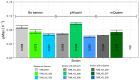
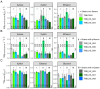
In Saccharomyces cerevisiae, pH homeostasis is reliant on ATP due to the use of proton-translocating ATPase (H+-ATPase) which constitutes a major drain within cellular ATP supply. Here, an exogenous proton-translocating pyrophosphatase (H+-PPase) from Arabidopsis thaliana, which uses inorganic pyrophosphate (PPi) rather than ATP, was evaluated for its effect on reducing the ATP burden. The H+-Ppase was localized to the vacuolar membrane or to the cell membrane, and their impact was studied under acetate stress at a low pH. Biosensors (pHluorin and mQueen-2m) were used to observe changes in intracellular pH (pHi) and ATP levels during growth on either glucose or xylose. A significant improvement of 35% in the growth rate at a pH of 3.7 and 6 g·L-1 acetic acid stress was observed in the vacuolar membrane H+-PPase strain compared to the parent strain. ATP levels were elevated in the same strain during anaerobic glucose and xylose fermentations. During anaerobic xylose fermentations, co-expression of pHluorin and a vacuolar membrane H+-PPase improved the growth characteristics by means of an improved growth rate (11.4%) and elongated logarithmic growth duration. Our study identified a potential method for improving productivity in the use of S. cerevisiae as a cell factory under the harsh conditions present in industry.
Keywords: ATP; Saccharomyces cerevisiae; acetic acid; glucose; mQueen-2m; pH homeostasis; pHluorin; proton translocating pyrophosphatase (H+-PPase); proton-translocating ATPase (H+-ATPase); xylose.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
High V-PPase activity is beneficial under high salt loads, but detrimental without salinity.New Phytol. 2018 Sep;219(4):1421-1432. doi: 10.1111/nph.15280. Epub 2018 Jun 25. New Phytol. 2018. PMID: 29938800 Free PMC article.
-
The flip side of the Arabidopsis type I proton-pumping pyrophosphatase (AVP1): Using a transmembrane H+ gradient to synthesize pyrophosphate.J Biol Chem. 2019 Jan 25;294(4):1290-1299. doi: 10.1074/jbc.RA118.006315. Epub 2018 Dec 3. J Biol Chem. 2019. PMID: 30510138 Free PMC article.
-
A plant proton-pumping inorganic pyrophosphatase functionally complements the vacuolar ATPase transport activity and confers bafilomycin resistance in yeast.Biochem J. 2011 Jul 15;437(2):269-78. doi: 10.1042/BJ20110447. Biochem J. 2011. PMID: 21612578
-
Vacuolar H(+)-translocating pyrophosphatases: a new category of ion translocase.Trends Biochem Sci. 1992 Sep;17(9):348-53. doi: 10.1016/0968-0004(92)90313-x. Trends Biochem Sci. 1992. PMID: 1329278 Review.
-
Biochemical, Structural and Physiological Characteristics of Vacuolar H+-Pyrophosphatase.Plant Cell Physiol. 2018 Jul 1;59(7):1300-1308. doi: 10.1093/pcp/pcy054. Plant Cell Physiol. 2018. PMID: 29534212 Review.
References
-
- Sahoo A., Das P.K., Patra S., Veeranki V.D. Advances in Yeast Biotechnology for Biofuels and Sustainability. Elsevier; Amsterdam, The Netherlands: 2023. Engineered Yeasts for the Production of Biofuel and Platform Chemicals; pp. 21–46.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

