Exosome-Mediated Antigen Delivery: Unveiling Novel Strategies in Viral Infection Control and Vaccine Design
- PMID: 38543914
- PMCID: PMC10974137
- DOI: 10.3390/vaccines12030280
Exosome-Mediated Antigen Delivery: Unveiling Novel Strategies in Viral Infection Control and Vaccine Design
Abstract
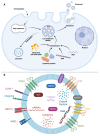
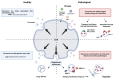
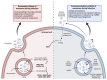
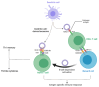
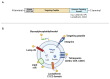
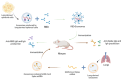
Exosomes are small subtypes of extracellular vesicles (EVs) naturally released by different types of cells into their environment. Their physiological roles appear to be multiple, yet many aspects of their biological activities remain to be understood. These vesicles can transport and deliver a variety of cargoes and may serve as unconventional secretory vesicles. Thus, they play a crucial role as important vectors for intercellular communication and the maintenance of homeostasis. Exosome production and content can vary under several stresses or modifications in the cell microenvironment, influencing cellular responses and stimulating immunity. During infectious processes, exosomes are described as double-edged swords, displaying both beneficial and detrimental effects. Owing to their tractability, the analysis of EVs from multiple biofluids has become a booming tool for monitoring various pathologies, from infectious to cancerous origins. In this review, we present an overview of exosome features and discuss their particular and ambiguous functions in infectious contexts. We then focus on their properties as diagnostic or therapeutic tools. In this regard, we explore the capacity of exosomes to vectorize immunogenic viral antigens and their function in mounting adaptive immune responses. As exosomes provide interesting platforms for antigen presentation, we further review the available data on exosome engineering, which enables peptides of interest to be exposed at their surface. In the light of all these data, exosomes are emerging as promising avenues for vaccine strategies.
Keywords: antigen presentation; cargoes; exosome engineering; exosomes; extracellular vesicles; flaviviruses; infection; pathological biomarkers; vaccine design; viruses.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

