Romosozumab added to ongoing denosumab in postmenopausal osteoporosis, a prospective observational study
- PMID: 38544922
- PMCID: PMC10972672
- DOI: 10.1093/jbmrpl/ziae016
Romosozumab added to ongoing denosumab in postmenopausal osteoporosis, a prospective observational study
Abstract
Background: Optimization of sequential and combination treatment is crucial in shaping long-term management of postmenopausal osteoporosis (OP).
Methods: We conducted a 6-month prospective observational study on postmenopausal women with severe OP receiving treatment with romosozumab either alone (in patients naïve to treatment) or in combination with ongoing long-term denosumab (>2 years) or continuing ongoing denosumab alone (>2 years). We collected serum samples for bone turnover markers, bone modulators, and calcium phosphate metabolism at baseline, month 3 and month 6. BMD was assessed at baseline and after 6 months.
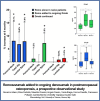
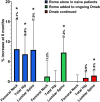
Results: Fifty-two postmenopausal women with OP were included in the study. Nineteen received romosozumab alone, 11 received romosozumab combined to ongoing denosumab, and 22 continued denosumab alone. BMD increased significantly at all sites at 6 months of follow-up in the romosozumab alone group (femoral neck +8.1%, total hip +6.8%, and lumbar spine +7.9%). In contrast, BMD increased significantly only at lumbar spine in the combination group (+7.2%) and in the denosumab group (+1.5%). P1nP increased significantly in romosozumab groups at month 3 (+70.4% in romosozumab alone group and +99.1% in combination group). Sclerostin levels increased steeply in both romosozumab groups, and Dkk1 did not change.
Conclusion: Romosozumab added to ongoing denosumab resulted in an increase in P1nP and lumbar spine BMD, but not in femoral neck BMD. For patients on denosumab, using romosozumab as an additional treatment appeared to be useful in terms of bone formation markers and spine BMD vs denosumab alone. Further randomized controlled trials, possibly powered to fracture outcomes, are needed to confirm our results.
Keywords: bone metabolism; bone mineral density (BMD); bone turnover markers (BTMs); denosumab; romosozumab.
© The Author(s) 2024. Published by Oxford University Press on behalf of the American Society for Bone and Mineral Research.
Conflict of interest statement
G.A. has received advisory board honoraria, consultancy fees, and/or speaker fees from Theramex, UCB, Lilly, Galapagos, Fresenius Kabi, Amgen, BMS, Abiogen, and Pfizer. D.G. has received advisory board honoraria, consultancy fees, and/or speaker fees from Abiogen, Celgene, Eli-Lilly, Neopharmed-Gentili, Pfizer, and UCB. M.R. reports advisory board honoraria, consultancy fees, and/or speaker fees from AbbVie, BMS, Eli-Lilly, Galapagos, Menarini, and UCB outside the submitted work. O.V. has received advisory board honoraria and speaker fees from Gilead, Fresenius Kabi, Biogen, Eli-Lilly, UCB, Abbvie, MSD, and BMS. A.F. reports personal fees from Abiogen, Novartis, and Neopharmed.
Figures




References
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

