Chronic nicotine exposure is associated with electrophysiological and sympathetic remodeling in the intact rabbit heart
- PMID: 38551482
- PMCID: PMC11381014
- DOI: 10.1152/ajpheart.00749.2023
Chronic nicotine exposure is associated with electrophysiological and sympathetic remodeling in the intact rabbit heart
Abstract
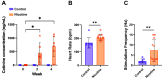
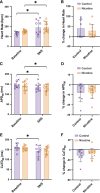
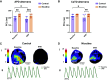
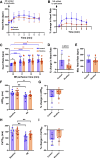
Nicotine is the primary addictive component of tobacco products. Through its actions on the heart and autonomic nervous system, nicotine exposure is associated with electrophysiological changes and increased arrhythmia susceptibility. To assess the underlying mechanisms, we treated rabbits with transdermal nicotine (NIC, 21 mg/day) or control (CT) patches for 28 days before performing dual optical mapping of transmembrane potential (RH237) and intracellular Ca2+ (Rhod-2 AM) in isolated hearts with intact sympathetic innervation. Sympathetic nerve stimulation (SNS) was performed at the first to third thoracic vertebrae, and β-adrenergic responsiveness was additionally evaluated following norepinephrine (NE) perfusion. Baseline ex vivo heart rate (HR) and SNS stimulation threshold were higher in NIC versus CT (P = 0.004 and P = 0.003, respectively). Action potential duration alternans emerged at longer pacing cycle lengths (PCL) in NIC versus CT at baseline (P = 0.002) and during SNS (P = 0.0003), with similar results obtained for Ca2+ transient alternans. SNS shortened the PCL at which alternans emerged in CT but not in NIC hearts. NIC-exposed hearts tended to have slower and reduced HR responses to NE perfusion, but ventricular responses to NE were comparable between groups. Although fibrosis was unaltered, NIC hearts had lower sympathetic nerve density (P = 0.03) but no difference in NE content versus CT. These results suggest both sympathetic hypoinnervation of the myocardium and regional differences in β-adrenergic responsiveness with NIC. This autonomic remodeling may contribute to the increased risk of arrhythmias associated with nicotine exposure, which may be further exacerbated with long-term use.NEW & NOTEWORTHY Here, we show that chronic nicotine exposure was associated with increased heart rate, increased susceptibility to alternans, and reduced sympathetic electrophysiological responses in the intact rabbit heart. We suggest that this was due to sympathetic hypoinnervation of the myocardium and diminished β-adrenergic responsiveness of the sinoatrial node following nicotine treatment. Though these differences did not result in increased arrhythmia propensity in our study, we hypothesize that prolonged nicotine exposure may exacerbate this proarrhythmic remodeling.
Keywords: action potential; arrhythmia; calcium; nicotine; sympathetic.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Crystal Ripplinger is an editor of
Figures


Update of
-
Chronic nicotine exposure is associated with electrophysiological and sympathetic remodeling in the intact rabbit heart.bioRxiv [Preprint]. 2023 Nov 23:2023.11.23.567754. doi: 10.1101/2023.11.23.567754. bioRxiv. 2023. Update in: Am J Physiol Heart Circ Physiol. 2024 Jun 1;326(6):H1337-H1349. doi: 10.1152/ajpheart.00749.2023. PMID: 38045290 Free PMC article. Updated. Preprint.
Comment in
-
Through the haze: unveiling the link between smoking and atrial fibrillation.Am J Physiol Heart Circ Physiol. 2024 Jun 1;326(6):H1489-H1490. doi: 10.1152/ajpheart.00286.2024. Epub 2024 May 17. Am J Physiol Heart Circ Physiol. 2024. PMID: 38758125 No abstract available.
Similar articles
-
Sympathetic structural and electrophysiological remodeling in a rabbit model of reperfused myocardial infarction.Am J Physiol Heart Circ Physiol. 2024 Sep 1;327(3):H631-H638. doi: 10.1152/ajpheart.00398.2024. Epub 2024 Jul 19. Am J Physiol Heart Circ Physiol. 2024. PMID: 39028283 Free PMC article.
-
Chronic nicotine exposure is associated with electrophysiological and sympathetic remodeling in the intact rabbit heart.bioRxiv [Preprint]. 2023 Nov 23:2023.11.23.567754. doi: 10.1101/2023.11.23.567754. bioRxiv. 2023. Update in: Am J Physiol Heart Circ Physiol. 2024 Jun 1;326(6):H1337-H1349. doi: 10.1152/ajpheart.00749.2023. PMID: 38045290 Free PMC article. Updated. Preprint.
-
Age-related changes in cardiac electrophysiology and calcium handling in response to sympathetic nerve stimulation.J Physiol. 2018 Sep;596(17):3977-3991. doi: 10.1113/JP276396. Epub 2018 Aug 3. J Physiol. 2018. PMID: 29938794 Free PMC article.
-
Molecular Mechanisms of Sympathetic Remodeling and Arrhythmias.Circ Arrhythm Electrophysiol. 2016 Feb;9(2):e001359. doi: 10.1161/CIRCEP.115.001359. Circ Arrhythm Electrophysiol. 2016. PMID: 26810594 Free PMC article. Review. No abstract available.
-
Addictive drugs, arrhythmias, and cardiac inward rectifiers.Europace. 2017 Mar 1;19(3):346-355. doi: 10.1093/europace/euw071. Europace. 2017. PMID: 27302393 Review.
Cited by
-
Sympathetic structural and electrophysiological remodeling in a rabbit model of reperfused myocardial infarction.Am J Physiol Heart Circ Physiol. 2024 Sep 1;327(3):H631-H638. doi: 10.1152/ajpheart.00398.2024. Epub 2024 Jul 19. Am J Physiol Heart Circ Physiol. 2024. PMID: 39028283 Free PMC article.
References
-
- Deo R, Norby FL, Katz R, Sotoodehnia N, Adabag S, DeFilippi CR, Kestenbaum B, Chen LY, Heckbert SR, Folsom AR, Kronmal RA, Konety S, Patton KK, Siscovick D, Shlipak MG, Alonso A. Development and validation of a sudden cardiac death prediction model for the general population. Circulation 134: 806–816, 2016. doi:10.1161/CIRCULATIONAHA.116.023042. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

