A real-world retrospective-prospective analysis of efficacy and safety of combined ixazomib, lenalidomide, and dexamethasone in relapsed/refractory multiple myeloma: The northern Italy experience
- PMID: 38558233
- PMCID: PMC10983806
- DOI: 10.1002/cam4.7071
A real-world retrospective-prospective analysis of efficacy and safety of combined ixazomib, lenalidomide, and dexamethasone in relapsed/refractory multiple myeloma: The northern Italy experience
Abstract
Introduction: Ixazomib, lenalidomide, and dexamethasone (IRd) have been approved for the treatment of relapsed/refractory multiple myeloma (RRMM) based on the results of the TOURMALINE-MM1.
Objectives and methods: We conducted a retrospective-prospective analysis of 106 RRMM patients (pts) treated with IRd in 21 centers in Northern Italy, with the aim to evaluate the efficacy and safety of IRd in real life.
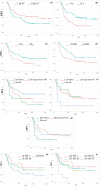
Results: At IRd initiation, 34% of pts were aged ≥75 (median 72.5), 8.5% had an ECOG performance status ≥2, 54.7% of evaluable pts carried high-risk cytogenetic abnormalities [del17p and/or t(4;14) and/or t(14;16) and/or 1 g gain/amp], 60.2% had received ≥2 prior lines of therapy (pLoT), 57.5% were lenalidomide (Len)-exposed (including both Len-sensitive and Len-refractory pts), and 22% were Len-refractory. Main G ≥3 adverse events (AEs) were thrombocytopenia (16%) and neutropenia (12.3%). G ≥3 non-hematologic AEs included infections (9.4%) and GI toxicity (diarrhea 5.7%, hepatotoxicity 2.8%), VTE, skin rash, and peripheral neuropathy were mainly G1-2. The overall response rate was 56.4% (≥VGPR 30%). With a median follow-up of 38 m, median PFS (mPFS) was 16 m and the 1-year OS rate was 73%. By subgroup analysis, an extended PFS was observed for pts achieving ≥VGPR (mPFS 21.2 m), time from diagnosis to IRd ≥5 years (26.2 m), 1 pLoT (34.4 m), Len-naïve (NR), age ≥70 (20 m). In pts exposed to Len, non-refractory in any prior line and immediately prior to IRd, mPFS was 16 and 18 m, respectively. An inferior PFS was seen in Len-refractory pts (4.6 m). By multivariate analysis, independent predictors of PFS were age ≥70 (HR 0.6), time from diagnosis ≥5 years (HR 0.32), refractoriness to Len in any prior line (HR 3.33), and immediately prior (HR 4.31).
Conclusion: IRd might be effective and safe in RRMM pts with an indolent disease, in early lines of treatment, and who proved Len-sensitive, independent of age, and cytogenetic risk.
Keywords: efficacy; ixazomib–lenalidomide–dexamethasone; lenalidomide‐exposed; lenalidomide‐refractory; real‐life; relapsed/refractory multiple myeloma; safety.
© 2024 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Leleu X, Masszi T, Bahlis NJ, et al. Patient‐reported health‐related quality of life from the phase III TOURMALINE‐MM1 study of ixazomib‐lenalidomide‐dexamethasone versus placebo‐lenalidomide‐dexamethasone in relapsed/refractory multiple myeloma. Am J Hematol. 2018;93:985‐993. doi:10.1002/ajh.25134 - DOI - PubMed
-
- Chari A, Romanus D, Palumbo A, et al. Randomized clinical trial representativeness and outcomes in real‐world patients: comparison of 6 Hallmark randomized clinical trials of relapsed/refractory multiple myeloma. Clin Lymphoma Myeloma Leuk. 2020;20(1):8‐17.e16. doi:10.1016/j.clml.2019.09.625 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

