Enhancing Cisplatin Efficacy with Low Toxicity in Solid Breast Cancer Cells Using pH-Charge-Reversal Sericin-Based Nanocarriers: Development, Characterization, and In Vitro Biological Assessment
- PMID: 38560009
- PMCID: PMC10976391
- DOI: 10.1021/acsomega.3c09361
Enhancing Cisplatin Efficacy with Low Toxicity in Solid Breast Cancer Cells Using pH-Charge-Reversal Sericin-Based Nanocarriers: Development, Characterization, and In Vitro Biological Assessment
Abstract
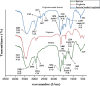
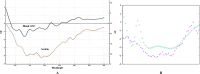
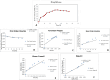
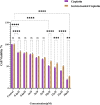
Platinum-based chemotherapeutic agents are widely employed in cancer treatment because of their effectiveness in targeting DNA. However, this indiscriminate action often affects both cancerous and normal cells, leading to severe side effects and highlighting the need for innovative approaches in achieving precise drug delivery. Nanotechnology presents a promising avenue for addressing these challenges. Protein-based nanocarriers exhibit promising capabilities in the realm of cancer drug delivery with silk sericin nanoparticles standing out as a leading contender. This investigation focuses on creating a sericin-based nanocarrier (SNC) featuring surface charge reversal designed to effectively transport cisplatin (Cispt-SNC) into MCF-7 breast cancer cells. Utilizing AutoDock4.2, our molecular docking analyses identified key amino acids and revealed distinctive conformational clusters, providing insights into the drug-protein interaction landscape and highlighting the potential of sericin as a carrier for controlled drug release. The careful optimization and fabrication of sericin as the carrier material were achieved through flash nanoprecipitation, a straightforward and reproducible method that is devoid of intricate equipment. The physicochemical properties of SNCs and Cispt-SNCs, particularly concerning size, surface charge, and morphology, were evaluated using dynamic light scattering (DLS) and scanning electron microscopy (SEM). Chemical and conformational analyses of the nanocarriers were conducted using Fourier-transform infrared spectroscopy (FTIR) and circular dichroism (CD), and elemental composition analysis was performed through energy-dispersive X-ray spectroscopy (EDX). This approach aimed to achieve the smallest nanoparticle size for Cispt-SNCs (180 nm) and high drug encapsulation efficiency (84%) at an optimal sericin concentration of 0.1% (w/v), maintaining a negative net charge at a physiological pH (7.4). Cellular uptake and cytotoxicity were investigated in MCF-7 breast cancer cells. SNCs demonstrated stability and exhibited a pH-dependent drug release behavior, aligning with the mildly acidic tumor microenvironment (pH 6.0-7.0). Efficient cellular uptake of Cispt-SNC, along with DNA fragmentation and chromatin condensation, was found at pH 6, leading to cell apoptosis. These results collectively indicate the potential of SNCs for achieving controlled drug release in a tumor-specific context. Our in vitro studies reveal the cytotoxicity of both cisplatin and Cispt-SNCs on MCF-7 cells. Cisplatin significantly reduced cell viability at 10 μM concentration (IC50), and the unique combination of sericin and cisplatin showcased enhanced cell viability compared to cisplatin alone, suggesting that controlled drug release is indicated by a gradient decrease in cell viability and highlighting SNCs as promising carriers. The study underscores the promise of protein-based nanocarriers in advancing targeted drug delivery for cancer therapy.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
A comparative study of sericin and gluten for magnetic nanoparticle-mediated drug delivery to breast cancer cell lines.Sci Rep. 2024 Aug 5;14(1):18150. doi: 10.1038/s41598-024-69009-y. Sci Rep. 2024. PMID: 39103485 Free PMC article.
-
Codelivery of resveratrol melatonin utilizing pH responsive sericin based nanocarriers inhibits the proliferation of breast cancer cell line at the different pH.Sci Rep. 2023 Jul 8;13(1):11090. doi: 10.1038/s41598-023-37668-y. Sci Rep. 2023. PMID: 37422485 Free PMC article.
-
In Vitro Interaction of Doxorubicin-Loaded Silk Sericin Nanocarriers with MCF-7 Breast Cancer Cells Leads to DNA Damage.Polymers (Basel). 2021 Jun 22;13(13):2047. doi: 10.3390/polym13132047. Polymers (Basel). 2021. PMID: 34206674 Free PMC article.
-
Advancing Nanomedicine Through Electron Microscopy: Insights Into Nanoparticle Cellular Interactions and Biomedical Applications.Int J Nanomedicine. 2025 Mar 8;20:2847-2878. doi: 10.2147/IJN.S500978. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40078651 Free PMC article. Review.
-
Hydrophobic amino acids grafted onto chitosan: a novel amphiphilic chitosan nanocarrier for hydrophobic drugs.Drug Dev Ind Pharm. 2017 Jan;43(1):1-11. doi: 10.1080/03639045.2016.1254240. Epub 2016 Nov 14. Drug Dev Ind Pharm. 2017. PMID: 27802776 Review.
Cited by
-
A comparative study of sericin and gluten for magnetic nanoparticle-mediated drug delivery to breast cancer cell lines.Sci Rep. 2024 Aug 5;14(1):18150. doi: 10.1038/s41598-024-69009-y. Sci Rep. 2024. PMID: 39103485 Free PMC article.
-
Sericin-coated MnO2@CeO2 nanocatalysts enable pH-responsive and synergistic vincristine delivery for lung cancer therapy.Sci Rep. 2025 Jul 11;15(1):25048. doi: 10.1038/s41598-025-10182-z. Sci Rep. 2025. PMID: 40646082 Free PMC article.
-
Recent Insights into the Potential and Challenges of Sericin as a Drug Delivery Platform for Multiple Biomedical Applications.Pharmaceutics. 2025 May 26;17(6):695. doi: 10.3390/pharmaceutics17060695. Pharmaceutics. 2025. PMID: 40574008 Free PMC article. Review.
References
-
- Becerra-Tomás N.; Balducci K.; Abar L.; Aune D.; Cariolou M.; Greenwood D. C.; Markozannes G.; Nanu N.; Vieira R.; Giovannucci E. L.; Gunter M. J.; Jackson A. A.; Kampman E.; Lund V.; Allen K.; Brockton N. T.; Croker H.; Katsikioti D.; McGinley-Gieser D.; Mitrou P.; Wiseman M.; Cross A. J.; Riboli E.; Clinton S. K.; McTiernan A.; Norat T.; Tsilidis K. K.; Chan D. S. M. Postdiagnosis dietary factors, supplement use and breast cancer prognosis: Global Cancer Update Programme (CUP Global) systematic literature review and meta-analysis. Int. J. Cancer 2023, 152 (4), 616–634. 10.1002/ijc.34321. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
