Biomimetic Chiral Nanomaterials with Selective Catalysis Activity
- PMID: 38561968
- PMCID: PMC11187969
- DOI: 10.1002/advs.202306979
Biomimetic Chiral Nanomaterials with Selective Catalysis Activity
Abstract
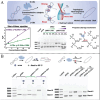
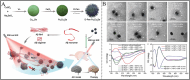
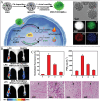
Chiral nanomaterials with unique chiral configurations and biocompatible ligands have been booming over the past decade for their interesting chiroptical effect, unique catalytical activity, and related bioapplications. The catalytic activity and selectivity of chiral nanomaterials have emerged as important topics, that can be potentially controlled and optimized by the rational biochemical design of nanomaterials. In this review, chiral nanomaterials synthesis, composition, and catalytic performances of different biohybrid chiral nanomaterials are discussed. The construction of chiral nanomaterials with multiscale chiral geometries along with the underlying principles for enhancing chiroptical responses are highlighted. Various biochemical approaches to regulate the selectivity and catalytic activity of chiral nanomaterials for biocatalysis are also summarized. Furthermore, attention is paid to specific chiral ligands, materials compositions, structure characteristics, and so on for introducing selective catalytic activities of representative chiral nanomaterials, with emphasis on substrates including small molecules, biological macromolecule, and in-site catalysis in living systems. Promising progress has also been emphasized in chiral nanomaterials featuring structural versatility and improved chiral responses that gave rise to unprecedented chances to utilize light for biocatalytic applications. In summary, the challenges, future trends, and prospects associated with chiral nanomaterials for catalysis are comprehensively proposed.
Keywords: biomimetic nanomaterials; chiroptical effects; selective catalysis activity.
© 2024 The Authors. Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures












References
-
- Ma W., Xu L., de Moura A. F., Wu X., Kuang H., Xu C., Kotov N. A., Chem. Rev. 2017, 117, 8041. - PubMed
-
- Xiao L., An T., Wang L., Xu X., Sun H., Nano Today 2020, 30, 100824.
-
- Im S. W., Ahn H. Y., Kim R. M., Cho N. H., Kim H., Lim Y. C., Lee H. E., Nam K. T., Adv. Mater. 2020, 32, 1905758.
-
- Zhu S., Sun M., Appl. Spectrosc. Rev. 2020, 56, 553.
-
- Fan J., Kotov N. A., Adv. Mater. 2020, 32, 1906738. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
