A flexible high-throughput cultivation protocol to assess the response of individuals' gut microbiota to diet-, drug-, and host-related factors
- PMID: 38562261
- PMCID: PMC10982853
- DOI: 10.1093/ismeco/ycae035
A flexible high-throughput cultivation protocol to assess the response of individuals' gut microbiota to diet-, drug-, and host-related factors
Abstract
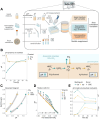
The anaerobic cultivation of fecal microbiota is a promising approach to investigating how gut microbial communities respond to specific intestinal conditions and perturbations. Here, we describe a flexible protocol using 96-deepwell plates to cultivate stool-derived gut microbiota. Our protocol aims to address gaps in high-throughput culturing in an anaerobic chamber. We characterized the influence of the gas phase on the medium chemistry and microbial physiology and introduced a modular medium preparation process to enable the testing of several conditions simultaneously. Furthermore, we identified a medium formulation that maximized the compositional similarity of ex vivo cultures and donor microbiota while limiting the bloom of Enterobacteriaceae. Lastly, we validated the protocol by demonstrating that cultivated fecal microbiota responded similarly to dietary fibers (resistant dextrin, soluble starch) and drugs (ciprofloxacin, 5-fluorouracil) as reported in vivo. This high-throughput cultivation protocol has the potential to facilitate culture-dependent studies, accelerate the discovery of gut microbiota-diet-drug-host interactions, and pave the way to personalized microbiota-centered interventions.
Keywords: 16S rRNA gene sequencing; anaerobic chamber; ex vivo; fecal microbiota; fibers; in vitro; microbial ecology; personalized microbiota response; short-chain fatty acids; xenobiotics.
© The Author(s) 2024. Published by Oxford University Press on behalf of the International Society for Microbial Ecology.
Conflict of interest statement
C.M., P.B., T.W., and G.L. are or were employees of PharmaBiome AG. T.W. and C.L. are founders of PharmaBiome AG. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Droplet-based high-throughput cultivation for accurate screening of antibiotic resistant gut microbes.Elife. 2020 Jun 17;9:e56998. doi: 10.7554/eLife.56998. Elife. 2020. PMID: 32553109 Free PMC article.
-
Validation of a batch cultivation protocol for fecal microbiota of Kenyan infants.BMC Microbiol. 2023 Jul 4;23(1):174. doi: 10.1186/s12866-023-02915-9. BMC Microbiol. 2023. PMID: 37403024 Free PMC article.
-
Fecal metabonomics combined with 16S rRNA gene sequencing to analyze the changes of gut microbiota in rats with kidney-yang deficiency syndrome and the intervention effect of You-gui pill.J Ethnopharmacol. 2019 Nov 15;244:112139. doi: 10.1016/j.jep.2019.112139. Epub 2019 Aug 8. J Ethnopharmacol. 2019. PMID: 31401318
-
The pig gut microbial diversity: Understanding the pig gut microbial ecology through the next generation high throughput sequencing.Vet Microbiol. 2015 Jun 12;177(3-4):242-51. doi: 10.1016/j.vetmic.2015.03.014. Epub 2015 Mar 23. Vet Microbiol. 2015. PMID: 25843944 Review.
-
Deciphering interactions between the gut microbiota and the immune system via microbial cultivation and minimal microbiomes.Immunol Rev. 2017 Sep;279(1):8-22. doi: 10.1111/imr.12578. Immunol Rev. 2017. PMID: 28856739 Free PMC article. Review.
Cited by
-
Cultivating efficiency: high-throughput growth analysis of anaerobic bacteria in compact microplate readers.Microbiol Spectr. 2024 May 2;12(5):e0365023. doi: 10.1128/spectrum.03650-23. Epub 2024 Mar 19. Microbiol Spectr. 2024. PMID: 38501820 Free PMC article.
-
Novel cross-feeding human gut microbes metabolizing tryptophan to indole-3-propionate.Gut Microbes. 2025 Dec;17(1):2501195. doi: 10.1080/19490976.2025.2501195. Epub 2025 May 7. Gut Microbes. 2025. PMID: 40336187 Free PMC article.
-
Cultivating efficiency: High-throughput growth analysis of anaerobic bacteria in compact microplate readers.bioRxiv [Preprint]. 2023 Oct 10:2023.10.10.561742. doi: 10.1101/2023.10.10.561742. bioRxiv. 2023. Update in: Microbiol Spectr. 2024 May 2;12(5):e0365023. doi: 10.1128/spectrum.03650-23. PMID: 37873238 Free PMC article. Updated. Preprint.
-
Deciphering oxidative stress responses in human gut microbes and fecal microbiota: a cultivation-based approach.FEMS Microbiol Ecol. 2025 May 20;101(6):fiaf054. doi: 10.1093/femsec/fiaf054. FEMS Microbiol Ecol. 2025. PMID: 40392681 Free PMC article.
-
Microbially-produced folate forms support the growth of Roseburia intestinalis but not its competitive fitness in fecal batch fermentations.BMC Microbiol. 2024 Sep 28;24(1):366. doi: 10.1186/s12866-024-03528-6. BMC Microbiol. 2024. PMID: 39342101 Free PMC article.
References
-
- Djekic D, Shi L, Brolin H et al. Effects of a vegetarian diet on cardiometabolic risk factors, gut microbiota, and plasma metabolome in subjects with ischemic heart disease: a randomized, crossover study. J Am Hear Assoc Cardiovasc Cerebrovasc Dis 2020;9:e016518. 10.1161/JAHA.120.016518. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

