Kinetics of Intermediate Release Enhances P450 11B2-Catalyzed Aldosterone Synthesis
- PMID: 38564530
- PMCID: PMC11259377
- DOI: 10.1021/acs.biochem.3c00725
Kinetics of Intermediate Release Enhances P450 11B2-Catalyzed Aldosterone Synthesis
Abstract
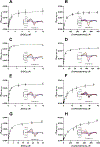
The mitochondrial enzyme cytochrome P450 11B2 (aldosterone synthase) catalyzes the 3 terminal transformations in the biosynthesis of aldosterone from 11-deoxycorticosterone (DOC): 11β-hydroxylation to corticosterone, 18-hydroxylation, and 18-oxidation. Prior studies have shown that P450 11B2 produces more aldosterone from DOC than from the intermediate corticosterone and that the reaction sequence is processive, with intermediates remaining bound to the active site between oxygenation reactions. In contrast, P450 11B1 (11β-hydroxylase), which catalyzes the terminal step in cortisol biosynthesis, shares a 93% amino acid sequence identity with P450 11B2, converts DOC to corticosterone, but cannot synthesize aldosterone from DOC. The biochemical and biophysical properties of P450 11B2, which enable its unique 18-oxygenation activity and processivity, yet are not also represented in P450 11B1, remain unknown. To understand the mechanism of aldosterone biosynthesis, we introduced point mutations at residue 320, which partially exchange the activities of P450 11B1 and P450 11B2 (V320A and A320V, respectively). We then investigated NADPH coupling efficiencies, binding kinetics and affinities, and product formation of purified P450 11B1 and P450 11B2, wild-type, and residue 320 mutations in phospholipid vesicles and nanodiscs. Coupling efficiencies for the 18-hydroxylase reaction with corticosterone as the substrate failed to correlate with aldosterone synthesis, ruling out uncoupling as a relevant mechanism. Conversely, corticosterone dissociation rates correlated inversely with aldosterone production. We conclude that intermediate dissociation kinetics, not coupling efficiency, enable P450 11B2 to synthesize aldosterone via a processive mechanism. Our kinetic data also suggest that the binding of DOC to P450 11B enzymes occurs in at least two distinct steps, favoring an induced-fit mechanism.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
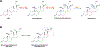
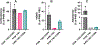
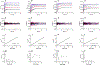
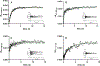
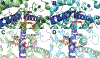
Similar articles
-
Human cytochrome P450 11B2 produces aldosterone by a processive mechanism due to the lactol form of the intermediate 18-hydroxycorticosterone.J Biol Chem. 2019 Aug 30;294(35):12975-12991. doi: 10.1074/jbc.RA119.009830. Epub 2019 Jul 11. J Biol Chem. 2019. PMID: 31296661 Free PMC article.
-
Molecular Recognition in Mitochondrial Cytochromes P450 That Catalyze the Terminal Steps of Corticosteroid Biosynthesis.Biochemistry. 2017 May 2;56(17):2282-2293. doi: 10.1021/acs.biochem.7b00034. Epub 2017 Apr 17. Biochemistry. 2017. PMID: 28355486 Free PMC article.
-
Catalytic modulation of human cytochromes P450 17A1 and P450 11B2 by phospholipid.J Steroid Biochem Mol Biol. 2018 Jul;181:63-72. doi: 10.1016/j.jsbmb.2018.03.003. Epub 2018 Mar 13. J Steroid Biochem Mol Biol. 2018. PMID: 29548669 Free PMC article.
-
Hypothesis: aldosterone is synthesized by an alternative pathway during severe sodium depletion. 'A new wine in an old bottle'.Clin Exp Pharmacol Physiol. 1998 May;25(5):369-78. doi: 10.1111/j.1440-1681.1998.tb02365.x. Clin Exp Pharmacol Physiol. 1998. PMID: 9612665 Review.
-
Disorders of the aldosterone synthase and steroid 11beta-hydroxylase deficiencies.Horm Res. 1999;51(5):211-22. doi: 10.1159/000023374. Horm Res. 1999. PMID: 10559665 Review.
Cited by
-
Updates on Mechanisms of Cytochrome P450 Catalysis of Complex Steroid Oxidations.Int J Mol Sci. 2024 Aug 20;25(16):9020. doi: 10.3390/ijms25169020. Int J Mol Sci. 2024. PMID: 39201706 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

