Cranial irradiation disrupts homeostatic microglial dynamic behavior
- PMID: 38570852
- PMCID: PMC10993621
- DOI: 10.1186/s12974-024-03073-z
Cranial irradiation disrupts homeostatic microglial dynamic behavior
Abstract
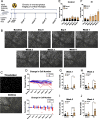
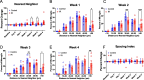
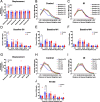
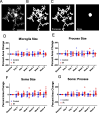
Cranial irradiation causes cognitive deficits that are in part mediated by microglia, the resident immune cells of the brain. Microglia are highly reactive, exhibiting changes in shape and morphology depending on the function they are performing. Additionally, microglia processes make dynamic, physical contacts with different components of their environment to monitor the functional state of the brain and promote plasticity. Though evidence suggests radiation perturbs homeostatic microglia functions, it is unknown how cranial irradiation impacts the dynamic behavior of microglia over time. Here, we paired in vivo two-photon microscopy with a transgenic mouse model that labels cortical microglia to follow these cells and determine how they change over time in cranial irradiated mice and their control littermates. We show that a single dose of 10 Gy cranial irradiation disrupts homeostatic cortical microglia dynamics during a 1-month time course. We found a lasting loss of microglial cells following cranial irradiation, coupled with a modest dysregulation of microglial soma displacement at earlier timepoints. The homogeneous distribution of microglia was maintained, suggesting microglia rearrange themselves to account for cell loss and maintain territorial organization following cranial irradiation. Furthermore, we found cranial irradiation reduced microglia coverage of the parenchyma and their surveillance capacity, without overtly changing morphology. Our results demonstrate that a single dose of radiation can induce changes in microglial behavior and function that could influence neurological health. These results set the foundation for future work examining how cranial irradiation impacts complex cellular dynamics in the brain which could contribute to the manifestation of cognitive deficits.
Keywords: Microglia; Radiation; Two-photon microscopy.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
In Vivo Imaging of the Microglial Landscape After Whole Brain Radiation Therapy.Int J Radiat Oncol Biol Phys. 2021 Nov 15;111(4):1066-1071. doi: 10.1016/j.ijrobp.2021.07.038. Epub 2021 Jul 24. Int J Radiat Oncol Biol Phys. 2021. PMID: 34314813 Free PMC article.
-
Cranial irradiation induces bone marrow-derived microglia in adult mouse brain tissue.J Radiat Res. 2014 Jul;55(4):713-9. doi: 10.1093/jrr/rru015. Epub 2014 Apr 4. J Radiat Res. 2014. PMID: 24706998 Free PMC article.
-
Aging-like changes in the transcriptome of irradiated microglia.Glia. 2015 May;63(5):754-67. doi: 10.1002/glia.22782. Epub 2015 Feb 17. Glia. 2015. PMID: 25690519 Free PMC article.
-
Cranial irradiation induces transient microglia accumulation, followed by long-lasting inflammation and loss of microglia.Oncotarget. 2016 Dec 13;7(50):82305-82323. doi: 10.18632/oncotarget.12929. Oncotarget. 2016. PMID: 27793054 Free PMC article.
-
Microglial Morphology Across Distantly Related Species: Phylogenetic, Environmental and Age Influences on Microglia Reactivity and Surveillance States.Front Immunol. 2021 Jun 18;12:683026. doi: 10.3389/fimmu.2021.683026. eCollection 2021. Front Immunol. 2021. PMID: 34220831 Free PMC article. Review.
Cited by
-
Cell-specific analysis of microglia following partial brain proton irradiation in mice.Clin Transl Radiat Oncol. 2025 Jun 27;54:101003. doi: 10.1016/j.ctro.2025.101003. eCollection 2025 Sep. Clin Transl Radiat Oncol. 2025. PMID: 40687733 Free PMC article.
-
The effects of P2Y12 loss on microglial gene expression, dynamics, and injury response in the cerebellum and cerebral cortex.Brain Behav Immun. 2025 Aug;128:99-120. doi: 10.1016/j.bbi.2025.03.036. Epub 2025 Mar 31. Brain Behav Immun. 2025. PMID: 40174868
-
Biological sex, microglial signaling pathways, and radiation exposure shape cortical proteomic profiles and behavior in mice.Brain Behav Immun Health. 2024 Nov 25;43:100911. doi: 10.1016/j.bbih.2024.100911. eCollection 2025 Feb. Brain Behav Immun Health. 2024. PMID: 39677060 Free PMC article.
-
Microglial dynamics and emerging therapeutic strategies in CNS homeostasis and pathology.Front Pharmacol. 2025 May 13;16:1577809. doi: 10.3389/fphar.2025.1577809. eCollection 2025. Front Pharmacol. 2025. PMID: 40432891 Free PMC article. Review.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

