Cell penetration of oxadiazole-containing macrocycles
- PMID: 38576720
- PMCID: PMC10989506
- DOI: 10.1039/d3cb00201b
Cell penetration of oxadiazole-containing macrocycles
Abstract
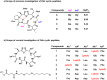
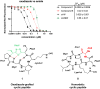
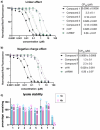
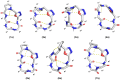
Passive membrane permeability is an important property in drug discovery and biological probe design. To elucidate the cell-penetrating ability of oxadiazole-containing (Odz) peptides, we employed the Chloroalkane Penetration Assay. The present study demonstrates that Odz cyclic peptides can be highly cell-penetrant depending on the position of specific side chains and the chloroalkane tag. Solution NMR shows that Odz cyclic peptides adopt a β-turn conformation. However, despite observing high cell penetration, we observed low passive permeability in experiments with artificial membranes. These findings highlight the complexity of controlling cell penetration for conformationally sensitive macrocycles and suggest that Odz cyclic peptides may provide a framework for designing cell-penetrant cyclic peptides.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources

