Upadacitinib for refractory ulcerative colitis with primary nonresponse to infliximab and vedolizumab: A case report
- PMID: 38576732
- PMCID: PMC10989418
- DOI: 10.12998/wjcc.v12.i9.1685
Upadacitinib for refractory ulcerative colitis with primary nonresponse to infliximab and vedolizumab: A case report
Abstract
Background: Many patients with ulcerative colitis (UC) do not respond well to, or tolerate conventional and biological therapies. There is currently no consensus on the treatment of refractory UC. Studies have demonstrated that the selective Janus kinase 1 inhibitor upadacitinib, a small-molecule drug, is effective and safe for treating UC. However, no studies have revealed that upadacitinib is effective in treating refractory UC with primary nonresponse to infliximab and vedolizumab.
Case summary: We report the case of a 44-year-old male patient with a chief complaint of bloody diarrhoea with mucus and pus, in addition to dizziness. The patient had recurrent disease after receiving mesalazine, prednisone, azathioprine, infliximab and vedolizumab over four years. Based on the endoscopic findings and pathological biopsy, the patient was diagnosed with refractory UC. In particular, the patient showed primary nonresponse to infliximab and vedolizumab. Based on the patient's history and recurrent disease, we decided to administer upadacitinib. During hospitalisation, the patient was received upadacitinib under our guidance. Eight weeks after the initiation of upadacitinib treatment, the patient's symptoms and endoscopic findings improved significantly. No notable adverse reactions have been reported to date.
Conclusion: Our case report suggests that upadacitinib may represent a valuable strategy for treating refractory UC with primary nonresponse.
Keywords: Case report; Infliximab; Primary nonresponse; Refractory ulcerative colitis; Upadacitinib; Vedolizumab.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors declare no conflict of interest.
Figures
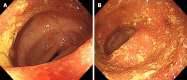


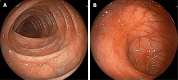
References
-
- Kobayashi T, Siegmund B, Le Berre C, Wei SC, Ferrante M, Shen B, Bernstein CN, Danese S, Peyrin-Biroulet L, Hibi T. Ulcerative colitis. Nat Rev Dis Primers. 2020;6:74. - PubMed
-
- Goetsch A, D'Amico F, Allocca M, Fiorino G, Furfaro F, Zilli A, Parigi TL, Radice S, Peyrin-Biroulet L, Danese S. Advances in pharmacotherapy for ulcerative colitis: A focus on JAK1 inhibitors. Expert Opin Pharmacother. 2023;24:849–861. - PubMed
-
- Danese S, Vermeire S, Zhou W, Pangan AL, Siffledeen J, Greenbloom S, Hébuterne X, D'Haens G, Nakase H, Panés J, Higgins PDR, Juillerat P, Lindsay JO, Loftus EV Jr, Sandborn WJ, Reinisch W, Chen MH, Sanchez Gonzalez Y, Huang B, Xie W, Liu J, Weinreich MA, Panaccione R. Upadacitinib as induction and maintenance therapy for moderately to severely active ulcerative colitis: Results from three phase 3, multicentre, double-blind, randomised trials. Lancet. 2022;399:2113–2128. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

