Safety and tolerability of topical trametinib in rosacea: Results from a phase I clinical trial
- PMID: 38577058
- PMCID: PMC10988662
- DOI: 10.1002/ski2.346
Safety and tolerability of topical trametinib in rosacea: Results from a phase I clinical trial
Abstract
Purpose: Overactivation of the mitogen activated kinase pathway has been associated with rosacea. We hypothesised that inhibitors of this pathway can be repurposed to alleviate rosacea symptoms.
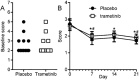
Methods: In order to test this hypothesis, we designed a double-blind, randomised, placebo-controlled phase I clinical trial to assess the safety and tolerability of a first-in-kind topical formulation of a MEK kinase inhibitor, trametinib. Subjects applied daily trametinib-containing cream (0.05 mg in 0.5 mL) to one cheek and cream without inhibitor to the other for consecutive 21 days. Skin irritation scores and blood samples were obtained during visits on days 8, 15 and 22.
Results: On analysis of high-performance liquid chromatography, no systemic trametinib absorption was detected during this treatment period. Subjects demonstrated a slight but significant improvement in both cheeks, regardless of drug contents. No adverse effects were reported during this time.
Conclusions: Topical trametinib was well tolerated at a dose of 0.05 mg per day without meaningful systemic absorption or local adverse events. A dose escalation trial is warranted to determine optimal dosing to treat rosacea while avoiding the adverse effects of systemic treatment.
© 2024 The Authors. Skin Health and Disease published by John Wiley & Sons Ltd on behalf of British Association of Dermatologists.
Conflict of interest statement
EJW and APA are co‐founders of Praxis Biotechnology, Inc. This company are inventors of and licensed an awarded patent (US‐20190142831‐A1) from Albany Medical College.
Figures
References
-
- Elewski BE. Results of a national rosacea patient survey: common issues that concern rosacea sufferers. J drugs dermatol JDD. 2009;8(2):120–123. - PubMed
LinkOut - more resources
Full Text Sources
