Quantitative and qualitative analysis of three DNA extraction methods from soybean, maize, and canola oils and investigation of the presence of genetically modified organisms (GMOs)
- PMID: 38577346
- PMCID: PMC10990853
- DOI: 10.1016/j.fochms.2024.100201
Quantitative and qualitative analysis of three DNA extraction methods from soybean, maize, and canola oils and investigation of the presence of genetically modified organisms (GMOs)
Abstract
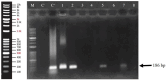
The objective of this study was to develop a DNA-based method for the identification and tracking of edible oils, which is important for health management. Three different DNA extraction methods (CTAB, MBST kit, and manual hexane-based method) were used to obtain high-purity DNA from crude and refined soybean, maize, and canola oils. PCR was then conducted using specific primers to identify the presence of genes related to each oil type and to assess transgenicity. The results showed that DNA was present in crude and refined oils, but in very low amounts. However, using method 3 for DNA extraction provided sufficient quantity and quality of DNA for successful PCR amplification. The study concluded that the main challenge in DNA extraction from oils is the presence of PCR inhibitors, which can be overcome using the manual hexane-based method. Also, the examination of protein presence in the oils using SDS-PAGE did not indicate any protein bands.
Keywords: Acetic acid glacial (PubChem CID: 176); CTAB (PubChem CID: 5974); Chloroform (PubChem CID: 6212); Coomassie brilliant blue G-250 (PubChem CID: 6324599); Crude and refined edible oils; DNA extraction; Ethanol (PubChem CID: 702); Genetically modified organisms (GMOs); Hexane (PubChem CID: 8058); Isopropanol (PubChem CID: 3776); Methanol (PubChem CID: 887); PCR inhibitors; Polymerase chain reaction (PCR).
© 2024 The Author(s).
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Development of nucleic acid isolation by non-silica-based nanoparticles and real-time PCR kit for edible vegetable oil traceability.Food Chem. 2019 Dec 1;300:125205. doi: 10.1016/j.foodchem.2019.125205. Epub 2019 Jul 18. Food Chem. 2019. PMID: 31330372
-
Production of three types of krill oils from krill meal by a three-step solvent extraction procedure.Food Chem. 2018 May 15;248:279-286. doi: 10.1016/j.foodchem.2017.12.068. Epub 2017 Dec 19. Food Chem. 2018. PMID: 29329855
-
Occurrence of deoxynivalenol in maize germs from North China Plain and the distribution of deoxynivalenol in the processed products of maize germs.Food Chem. 2018 Nov 15;266:557-562. doi: 10.1016/j.foodchem.2018.05.111. Epub 2018 May 25. Food Chem. 2018. PMID: 30381225
-
Natural volatile oils derived from herbal medicines: A promising therapy way for treating depressive disorder.Pharmacol Res. 2021 Feb;164:105376. doi: 10.1016/j.phrs.2020.105376. Epub 2020 Dec 11. Pharmacol Res. 2021. PMID: 33316383 Review.
-
The effects of the phytochemistry of cocoa on the food chemistry of chocolate(s) and how disease resistance in cocoa can be improved using CRISPR/Cas9 technology.Food Chem (Oxf). 2021 Sep 22;3:100043. doi: 10.1016/j.fochms.2021.100043. eCollection 2021 Dec 30. Food Chem (Oxf). 2021. PMID: 35415660 Free PMC article. Review.
Cited by
-
Novel PCR-Based Technology for the Detection of Sunflower in Edible and Used Cooking Oils.Foods. 2024 Nov 24;13(23):3760. doi: 10.3390/foods13233760. Foods. 2024. PMID: 39682833 Free PMC article.
References
-
- Bessetti J. An introduction to PCR inhibitors. Journal of Microbiological Methods. 2007;28:159–167.
-
- Bogani P., Minunni M., Spiriti M.M., Zavaglia M., Tombelli S., Buiatti M., Mascini M. Transgenes monitoring in an industrial soybean processing chain by DNA-based conventional approaches and biosensors. Food Chemistry. 2009;113(2):658–664.
-
- Bosch A., Sánchez G., Abbaszadegan M., Carducci A., Guix S., Le Guyader F., Netshikweta R., Pintó R., Van der Poel W.H.M., Rutjes S., Sano D., Taylor M.B., Van Zyl W.B., Rodríguez-Lázaro D., Kovac K., Sellwood J. Analytical methods for virus detection in water and food. Food Analytical Methods. 2011;4(1):4–12.
-
- Bradford K.J., Van Deynze A., Gutterson N., Parrott W., Strauss S.H. Regulating transgenic crops sensibly: Lessons from plant breeding, biotechnology and genomics. Nature biotechnology. 2005;23(4):439–444. - PubMed
LinkOut - more resources
Full Text Sources

