Reactive Microneedle Patches with Antibacterial and Dead Bacteria-Trapping Abilities for Skin Infection Treatment
- PMID: 38582511
- PMCID: PMC11186059
- DOI: 10.1002/advs.202309622
Reactive Microneedle Patches with Antibacterial and Dead Bacteria-Trapping Abilities for Skin Infection Treatment
Abstract
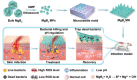
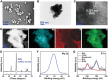
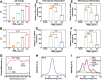
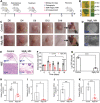
Bacterial skin infections are highly prevalent and pose a significant public health threat. Current strategies are primarily focused on the inhibition of bacterial activation while disregarding the excessive inflammation induced by dead bacteria remaining in the body and the effect of the acidic microenvironment during therapy. In this study, a novel dual-functional MgB2 microparticles integrated microneedle (MgB2 MN) patch is presented to kill bacteria and eliminate dead bacteria for skin infection management. The MgB2 microparticles not only can produce a local alkaline microenvironment to promote the proliferation and migration of fibroblasts and keratinocytes, but also achieve >5 log bacterial inactivation. Besides, the MgB2 microparticles effectively mitigate dead bacteria-induced inflammation through interaction with lipopolysaccharide (LPS). With the incorporation of these MgB2 microparticles, the resultant MgB2 MN patches effectively kill bacteria and capture dead bacteria, thereby mitigating these bacteria-induced inflammation. Therefore, the MgB2 MN patches show good therapeutic efficacy in managing animal bacterial skin infections, including abscesses and wounds. These results indicate that reactive metal borides-integrated microneedle patches hold great promise for the treatment of clinical skin infections.
Keywords: eliminate dead bacteria; microenvironment; microneedle; reactive metal boride; skin infection.
© 2024 The Authors. Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
It Takes Two to Tangle: Microneedle Patches Co-delivering Monoclonal Antibodies and Engineered Antimicrobial Peptides Effectively Eradicate Wound Biofilms.Macromol Biosci. 2024 May;24(5):e2300519. doi: 10.1002/mabi.202300519. Epub 2024 Jan 20. Macromol Biosci. 2024. PMID: 38217528
-
Flexible and Stretchable Microneedle Patches with Integrated Rigid Stainless Steel Microneedles for Transdermal Biointerfacing.PLoS One. 2016 Dec 9;11(12):e0166330. doi: 10.1371/journal.pone.0166330. eCollection 2016. PLoS One. 2016. PMID: 27935976 Free PMC article.
-
Development of a novel therapeutic approach using a retinoic acid-loaded microneedle patch for seborrheic keratosis treatment and safety study in humans.J Control Release. 2013 Oct 28;171(2):93-103. doi: 10.1016/j.jconrel.2013.06.008. Epub 2013 Jun 18. J Control Release. 2013. PMID: 23791975 Clinical Trial.
-
Microneedle patches for vaccination in developing countries.J Control Release. 2016 Oct 28;240:135-141. doi: 10.1016/j.jconrel.2015.11.019. Epub 2015 Nov 18. J Control Release. 2016. PMID: 26603347 Free PMC article. Review.
-
Advances in Antimicrobial Microneedle Patches for Combating Infections.Adv Mater. 2020 Aug;32(33):e2002129. doi: 10.1002/adma.202002129. Epub 2020 Jun 29. Adv Mater. 2020. PMID: 32602146 Review.
Cited by
-
Engineered Microneedle System Enables the Smart Regulation of Nanodynamic Sterilization and Tissue Regeneration for Wound Management.Adv Sci (Weinh). 2025 Mar;12(9):e2412226. doi: 10.1002/advs.202412226. Epub 2025 Jan 13. Adv Sci (Weinh). 2025. PMID: 39804981 Free PMC article.
-
Nature-derived microneedles with metal-polyphenolic networks encapsulation for chronic soft tissue defects repair: Responding and remodeling the regenerative microenvironment.Mater Today Bio. 2025 Feb 1;31:101539. doi: 10.1016/j.mtbio.2025.101539. eCollection 2025 Apr. Mater Today Bio. 2025. PMID: 40026624 Free PMC article.
-
Living photosynthetic microneedle patches for in situ oxygenation and postsurgical melanoma therapy.J Nanobiotechnology. 2024 Nov 11;22(1):698. doi: 10.1186/s12951-024-02982-8. J Nanobiotechnology. 2024. PMID: 39529107 Free PMC article.
-
Microneedles as transdermal drug delivery system for enhancing skin disease treatment.Acta Pharm Sin B. 2024 Dec;14(12):5161-5180. doi: 10.1016/j.apsb.2024.08.013. Epub 2024 Aug 22. Acta Pharm Sin B. 2024. PMID: 39807331 Free PMC article. Review.
-
Programmed Transformation of Osteogenesis Microenvironment by a Multifunctional Hydrogel to Enhance Repair of Infectious Bone Defects.Adv Sci (Weinh). 2025 Mar;12(10):e2409683. doi: 10.1002/advs.202409683. Epub 2025 Jan 22. Adv Sci (Weinh). 2025. PMID: 39840502 Free PMC article.
References
-
- Raff A. B., Kroshinsky D., J. Am. Med. Assoc. 2016, 316, 325.
-
- Choi V., Rohn J. L., Stoodley P., Carugo D., Stride E., Nat. Rev. Microbiol. 2023, 21, 555. - PubMed
MeSH terms
Substances
Grants and funding
- 82102335/National Natural Science Foundation of China
- 52073060/National Natural Science Foundation of China
- 2020YFA0908200/National Key Research and Development Program of China
- 2021B1515120054/Guangdong Basic and Applied Basic Research Foundation
- JCYJ20190813152616459/Shenzhen Fundamental Research Program
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
