Synthesis, anticancer evaluation, molecular docking and ADME study of novel pyrido[4',3':3,4]pyrazolo[1,5-a]pyrimidines as potential tropomyosin receptor kinase A (TrKA) inhibitors
- PMID: 38582910
- PMCID: PMC10999085
- DOI: 10.1186/s13065-024-01166-7
Synthesis, anticancer evaluation, molecular docking and ADME study of novel pyrido[4',3':3,4]pyrazolo[1,5-a]pyrimidines as potential tropomyosin receptor kinase A (TrKA) inhibitors
Abstract
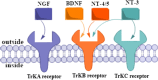
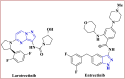
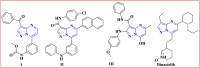
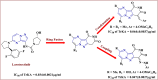
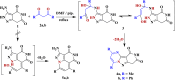
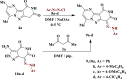
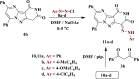
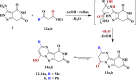
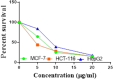
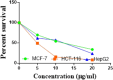
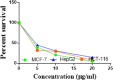
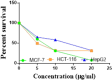
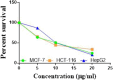
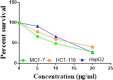
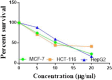
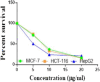
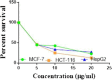
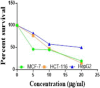
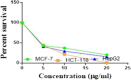
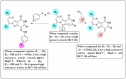
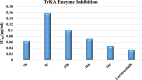
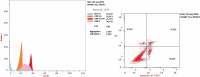
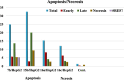
The starting compound 3-amino-1,7-dihydro-4H-pyrazolo[4,3-c]pyridine-4,6(5H)-dione (1) is reacted with each of diketone and β-ketoester, forming pyridopyrazolo[1,5-a]pyrimidines 4a,b and 14a,b, respectively. The compounds 4 and 14 reacted with each of aromatic aldehyde and arenediazonium salt to give the respective arylidenes and arylhydrazo derivatives, respectively. The structure of the new products was established using spectroscopic techniques. The cytotoxic activity of selected targets was tested in vitro against three cancer cell lines MCF7, HepG2 and HCT116. The data obtained from enzymatic assays of TrKA indicated that compounds 7b and 16c have the strongest inhibitory effects on TrKA with IC50 = 0.064 ± 0.0037 μg/ml and IC50 = 0.047 ± 0.0027 μg/ml, respectively, compared to the standard drug Larotrectinib with IC50 = 0.034 ± 0.0021 μg/ml for the HepG2 cancer cell line. In cell cycle analysis, compounds 7b, 15b, 16a and 16c caused the greatest arrest in cell cycle at the G2/M phase. In addition, compound 15b has a higher apoptosis-inducing effect (36.72%) than compounds 7b (34.70%), 16a (21.14) and 16c (26.54%). Compounds 7b, 16a and 16c were shown fit tightly into the active site of the TrKA kinase crystal structure (PDB: 5H3Q). Also, ADME study was performed on some selected potent anticancer compounds described in this study.
Keywords: ADME studies; Anticancer activity; Apoptotic activity; Molecular docking; Pyrido[4ʹ,3ʹ:3,4]pyrazolo[1,5-a]pyrimidines; TrKA enzyme.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



















References
-
- Yakes FM, Chen J, Tan J, Yamaguchi K, Shi Y, Yu P, Qian F, Chu F, Bentzien F, Cancilla B, Orf J, You A, Laird AD, Engst S, Lee L, Lesch J, Chou YC, Joly AH. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther. 2011;10:2298–2308. doi: 10.1158/1535-7163.MCT-11-0264. - DOI - PubMed
-
- Patwardhan PP, Ivy KS, Musi E, de Stanchina E, Schwartz GK. Significant blockade of multiple receptor tyrosine kinases by MGCD516 (Sitravatinib), a novel small molecule inhibitor, shows potent anti-tumor activity in preclinical models of sarcoma. Oncotarget. 2016;7:4093–4109. doi: 10.18632/oncotarget.6547. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
