Ligand and Linkage Isomers of Bis(ethylthiocarbamato) Copper Complexes with Cyclic C6H8 Backbone Substituents: Synthesis, Characterization, and Antiproliferation Activity
- PMID: 38584911
- PMCID: PMC10997340
- DOI: 10.1002/ejic.202300447
Ligand and Linkage Isomers of Bis(ethylthiocarbamato) Copper Complexes with Cyclic C6H8 Backbone Substituents: Synthesis, Characterization, and Antiproliferation Activity
Abstract
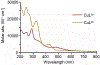
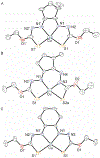
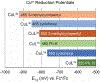
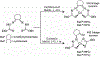
A series of isomeric bis(alkylthiocarbamate) copper complexes have been synthesized, characterized, and evaluated for antiproliferation activity. The complexes were derived from ligand isomers with 3-methylpentyl (H2L2) and cyclohexyl (H2L3) backbone substituents, which each yield a pair of linkage isomers. The thermodynamic products CuL2a/3a have two imino N and two S donors resulting in three five-member chelate rings (555 isomers). The kinetic isomers CuL2b/3b have one imino and one hydrazino N donor and two S donors resulting in four-, six-, and five-member rings (465 isomers). The 555 isomers have more accessible CuII/I potentials (E1/2 = -811/-768 mV vs. ferrocenium/ferrocene) and lower energy charge transfer bands than their 465 counterparts (E1/2 = -923/-854 mV). Antiproliferation activities were evaluated against the lung adenocarcinoma cell line (A549) and nonmalignant lung fibroblast cell line (IMR-90) using the MTT assay. CuL2a was potent (A549EC50 = 0.080 μM) and selective (IMR-90EC50/A549EC50 = 25) for A549. Its linkage isomer CuL2b had equivalent A549 activity, but lower selectivity (IMR-90EC50/A549EC50 = 12.5). The isomers CuL3a and CuL3b were less potent with A549EC50 values of 1.9 and 0.19 μM and less selective with IMR-90EC50/A549EC50 ratios of 2.3 and 2.65, respectively. There was no correlation between reduction potential and A549 antiproliferation activity/selectivity.
Keywords: N2S2 ligands; antiproliferation activity; copper; ligand isomer; linkage isomer.
Conflict of interest statement
The authors declare the following competing financial interest(s): Complexes described in this manuscript are included in an issued US patent (US-11208379-B2) entitled “Compounds, Compositions, Methods for Treating Diseases, and Methods for Preparing Compounds.” A portion of the results described in this manuscript are included in the dissertation Alkylthiocarbamate metal complexes with antiproliferation activity by Kritika Bajaj, University of Louisville.[21]
Figures
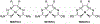

Similar articles
-
Physical structure of constitutional isomers influences antiproliferation activity of thiosemicarbazone-alkylthiocarbamate copper complexes.J Inorg Biochem. 2023 Sep;246:112288. doi: 10.1016/j.jinorgbio.2023.112288. Epub 2023 Jun 7. J Inorg Biochem. 2023. PMID: 37320890
-
Investigations of Bis(alkylthiocarbamato)copper Linkage Isomers.Inorg Chem. 2022 May 23;61(20):7715-7719. doi: 10.1021/acs.inorgchem.2c00371. Epub 2022 May 12. Inorg Chem. 2022. PMID: 35549215
-
Synthesis, Characterization, and Biological Activity of Hybrid Thiosemicarbazone-Alkylthiocarbamate Metal Complexes.Inorg Chem. 2020 Apr 6;59(7):4924-4935. doi: 10.1021/acs.inorgchem.0c00182. Epub 2020 Mar 11. Inorg Chem. 2020. PMID: 32159342
-
Great structural variety of complexes in copper(II)-oligoglycine systems: microspeciation and coordination modes as studied by the two-dimensional simulation of electron paramagnetic resonance spectra.J Am Chem Soc. 2003 Apr 30;125(17):5227-35. doi: 10.1021/ja021245+. J Am Chem Soc. 2003. PMID: 12708876
-
Unravelling the kinetics of electro- and photochemical S → O linkage isomerization in Ru(II)-NHC-DMSO complexes utilised for photoinduced substitution reactions.Dalton Trans. 2024 Jul 30;53(30):12662-12675. doi: 10.1039/d4dt01200c. Dalton Trans. 2024. PMID: 39012321
References
-
- Werner A, Ber. Dtsch. Chem. Ges 1907, 40, 765–788.
-
- Balahura RJ, Lewis NA, Coord. Chem. Rev 1976, 20, 109–153.
-
- Appleton TG, Coord. Chem. Rev 1997, 166, 313–359.
-
- Hazari A, Ghosh A, J. Indian Chem. Soc 2018, 95, 1597–1606.
-
- Aly MM, Stephanos JJ, Spectrochim. Acta A 1994, 50, 835–840;
- Lacey MJ, Macdonald C, Shannon J, Aust. J. Chem 1973, 26, 263–268.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
