Local contribution to the somatosensory evoked potentials in rat's thalamus
- PMID: 38593141
- PMCID: PMC11003638
- DOI: 10.1371/journal.pone.0301713
Local contribution to the somatosensory evoked potentials in rat's thalamus
Abstract
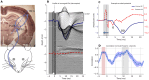
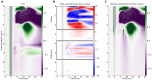
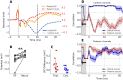
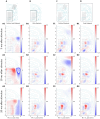
Local Field Potential (LFP), despite its name, often reflects remote activity. Depending on the orientation and synchrony of their sources, both oscillations and more complex waves may passively spread in brain tissue over long distances and be falsely interpreted as local activity at such distant recording sites. Here we show that the whisker-evoked potentials in the thalamic nuclei are of local origin up to around 6 ms post stimulus, but the later (7-15 ms) wave is overshadowed by a negative component reaching from cortex. This component can be analytically removed and local thalamic LFP can be recovered reliably using Current Source Density analysis. We used model-based kernel CSD (kCSD) method which allowed us to study the contribution of local and distant currents to LFP from rat thalamic nuclei and barrel cortex recorded with multiple, non-linear and non-regular multichannel probes. Importantly, we verified that concurrent recordings from the cortex are not essential for reliable thalamic CSD estimation. The proposed framework can be used to analyze LFP from other brain areas and has consequences for general LFP interpretation and analysis.
Copyright: © 2024 Średniawa et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Local field potentials and the encoding of whisker deflections by population firing synchrony in thalamic barreloids.J Neurophysiol. 2003 Apr;89(4):2137-45. doi: 10.1152/jn.00582.2002. Epub 2002 Dec 27. J Neurophysiol. 2003. PMID: 12612019
-
The neurogenesis of P1 and N1: A concurrent EEG/LFP study.Neuroimage. 2017 Feb 1;146:575-588. doi: 10.1016/j.neuroimage.2016.09.034. Epub 2016 Sep 16. Neuroimage. 2017. PMID: 27646129 Free PMC article.
-
Cross-trial correlation analysis of evoked potentials reveals arousal-related attenuation of thalamo-cortical coupling.J Comput Neurosci. 2010 Dec;29(3):485-93. doi: 10.1007/s10827-010-0220-0. Epub 2010 Feb 23. J Comput Neurosci. 2010. PMID: 20177762
-
Circuit dynamics and coding strategies in rodent somatosensory cortex.J Neurophysiol. 2000 Mar;83(3):1158-66. doi: 10.1152/jn.2000.83.3.1158. J Neurophysiol. 2000. PMID: 10712446
-
Is there a thalamic component to experience-dependent cortical plasticity?Philos Trans R Soc Lond B Biol Sci. 2002 Dec 29;357(1428):1709-15. doi: 10.1098/rstb.2002.1169. Philos Trans R Soc Lond B Biol Sci. 2002. PMID: 12626005 Free PMC article. Review.
Cited by
-
Deciphering Authentic Nociceptive Thalamic Responses in Rats.Research (Wash D C). 2024 Apr 9;7:0348. doi: 10.34133/research.0348. eCollection 2024. Research (Wash D C). 2024. PMID: 38617991 Free PMC article.
References
-
- Nunez PL, Srinivasan R. Electric Fields of the Brain. Oxford University Press; 2006.
MeSH terms
LinkOut - more resources
Full Text Sources

