The E3 ubiquitin ligase ITCH negatively regulates intercellular communication via gap junctions by targeting connexin43 for lysosomal degradation
- PMID: 38597989
- PMCID: PMC11006747
- DOI: 10.1007/s00018-024-05165-8
The E3 ubiquitin ligase ITCH negatively regulates intercellular communication via gap junctions by targeting connexin43 for lysosomal degradation
Abstract
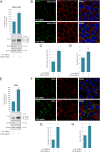
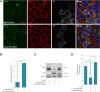
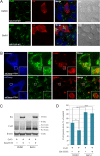
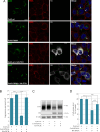
Intercellular communication via gap junctions has a fundamental role in regulating cell growth and tissue homeostasis, and its dysregulation may be involved in cancer development and radio- and chemotherapy resistance. Connexin43 (Cx43) is the most ubiquitously expressed gap junction channel protein in human tissues. Emerging evidence indicates that dysregulation of the sorting of Cx43 to lysosomes is important in mediating the loss of Cx43-based gap junctions in cancer cells. However, the molecular basis underlying this process is currently poorly understood. Here, we identified the E3 ubiquitin ligase ITCH as a novel regulator of intercellular communication via gap junctions. We demonstrate that ITCH promotes loss of gap junctions in cervical cancer cells, which is associated with increased degradation of Cx43 in lysosomes. The data further indicate that ITCH interacts with and regulates Cx43 ubiquitination and that the ITCH-induced loss of Cx43-based gap junctions requires its catalytic HECT (homologous to E6-AP C-terminus) domain. The data also suggest that the ability of ITCH to efficiently promote loss of Cx43-based gap junctions and degradation of Cx43 depends on a functional PY (PPXY) motif in the C-terminal tail of Cx43. Together, these data provide new insights into the molecular basis underlying the degradation of Cx43 and have implications for the understanding of how intercellular communication via gap junctions is lost during cancer development.
Keywords: Cervix; Connexin; Deubiquitination; Endosome; NEDD4.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests concerning this study.
Figures



References
-
- Mesnil M, Aasen T, Boucher J, Chepied A, Cronier L, Defamie N, Kameritsch P, Laird DW, Lampe PD, Lathia JD, Leithe E, Mehta PP, Monvoisin A, Pogoda K, Sin WC, Tabernero A, Yamasaki H, Yeh ES, Dagli MLZ, Naus CC. An update on minding the gap in cancer. Biochim Biophys Acta. 2018;1860:237–243. doi: 10.1016/j.bbamem.2017.06.015. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

