Engineering approaches to manipulate osteoclast behavior for bone regeneration
- PMID: 38600918
- PMCID: PMC11004223
- DOI: 10.1016/j.mtbio.2024.101043
Engineering approaches to manipulate osteoclast behavior for bone regeneration
Abstract
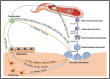
Extensive research has delved into the multifaceted roles of osteoclasts beyond their traditional function in bone resorption in recent years, uncovering their significant influence on bone formation. This shift in understanding has spurred investigations into engineering strategies aimed at leveraging osteoclasts to not only inhibit bone resorption but also facilitate bone regeneration. This review seeks to comprehensively examine the mechanisms by which osteoclasts impact bone metabolism. Additionally, it explores various engineering methodologies, including the modification of bioactive material properties, localized drug delivery, and the introduction of exogenous cells, assessing their potential and mechanisms in aiding bone repair by targeting osteoclasts. Finally, the review proposes current limitations and future routes for manipulating osteoclasts through biological and material cues to facilitate bone repair.
Keywords: Biomaterials; Bone coupling; Bone regeneration; Drug delivery; Osteoclasts.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Biomimetic polydopamine-laced hydroxyapatite collagen material orients osteoclast behavior to an anti-resorptive pattern without compromising osteoclasts' coupling to osteoblasts.Biomater Sci. 2021 Nov 9;9(22):7565-7574. doi: 10.1039/d1bm01119g. Biomater Sci. 2021. PMID: 34664567 Free PMC article.
-
The role of osteoclasts in bone tissue engineering.J Tissue Eng Regen Med. 2015 Oct;9(10):1133-49. doi: 10.1002/term.1851. Epub 2014 Jan 29. J Tissue Eng Regen Med. 2015. PMID: 24478169 Review.
-
Bioactives and Biomaterial Construction for Modulating Osteoclast Activities.Adv Healthc Mater. 2024 Mar;13(6):e2302807. doi: 10.1002/adhm.202302807. Epub 2023 Dec 7. Adv Healthc Mater. 2024. PMID: 38009952 Review.
-
Osteoclast-Driven Osteogenesis, Bone Remodeling and Biomaterial Resorption: A New Profile of BMP2-CPC-Induced Alveolar Bone Regeneration.Int J Mol Sci. 2022 Oct 13;23(20):12204. doi: 10.3390/ijms232012204. Int J Mol Sci. 2022. PMID: 36293100 Free PMC article.
-
Bioactive glass-ceramic for bone tissue engineering: an in vitro and in vivo study focusing on osteoclasts.Braz Oral Res. 2022 Mar 14;36:e022. doi: 10.1590/1807-3107bor-2022.vol36.0022. eCollection 2022. Braz Oral Res. 2022. PMID: 35293496
Cited by
-
Biomaterial Cues for Regulation of Osteoclast Differentiation and Function in Bone Regeneration.Adv Ther (Weinh). 2025 Jan;8(1):2400296. doi: 10.1002/adtp.202400296. Epub 2024 Nov 15. Adv Ther (Weinh). 2025. PMID: 39867107
-
Biodegradable Zn-0.8Mg-0.2Sr alloy as an internal fixation material exhibits controlled degradation with enhanced osteogenesis.RSC Adv. 2025 Aug 22;15(37):30071-30088. doi: 10.1039/d5ra02009c. eCollection 2025 Aug 22. RSC Adv. 2025. PMID: 40860423 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

