Neuroprotective effects of lactate and ketone bodies in acute brain injury
- PMID: 38603600
- PMCID: PMC11179615
- DOI: 10.1177/0271678X241245486
Neuroprotective effects of lactate and ketone bodies in acute brain injury
Abstract
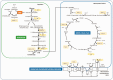
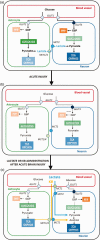
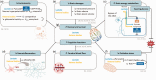
The goal of neurocritical care is to prevent and reverse the pathologic cascades of secondary brain injury by optimizing cerebral blood flow, oxygen supply and substrate delivery. While glucose is an essential energetic substrate for the brain, we frequently observe a strong decrease in glucose delivery and/or a glucose metabolic dysregulation following acute brain injury. In parallel, during the last decades, lactate and ketone bodies have been identified as potential alternative fuels to provide energy to the brain, both under physiological conditions and in case of glucose shortage. They are now viewed as integral parts of brain metabolism. In addition to their energetic role, experimental evidence also supports their neuroprotective properties after acute brain injury, regulating in particular intracranial pressure control, decreasing ischemic volume, and leading to an improvement in cognitive functions as well as survival. In this review, we present preclinical and clinical evidence exploring the mechanisms underlying their neuroprotective effects and identify research priorities for promoting lactate and ketone bodies use in brain injury.
Keywords: Alternative brain fuels; TBI; brain metabolism; hypoxia; ketone bodies; lactate; neuroprotection; stroke.
Conflict of interest statement
Declaration of conflicting interestsThe author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
References
-
- Patet C, Quintard H, Suys T, et al.. Neuroenergetic response to prolonged cerebral glucose depletion after severe brain injury and the role of lactate. J Neurotrauma 2015; 32: 1560–1566. - PubMed
-
- Swanson RA, Sagar SM, Sharp FR. Regional brain glycogen stores and metabolism during complete global ischaemia. Neurol Res 1989; 11: 24–28. - PubMed
-
- Vespa P, McArthur DL, Stein N, et al.. Tight glycemic control increases metabolic distress in traumatic brain injury: a randomized controlled within-subjects trial. Crit Care Med 2012; 40: 1923–1929. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

