Design and characterization of protective pan-ebolavirus and pan-filovirus bispecific antibodies
- PMID: 38603762
- PMCID: PMC11037526
- DOI: 10.1371/journal.ppat.1012134
Design and characterization of protective pan-ebolavirus and pan-filovirus bispecific antibodies
Abstract
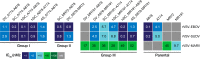
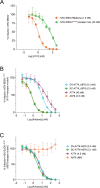
Monoclonal antibodies (mAbs) are an important class of antiviral therapeutics. MAbs are highly selective, well tolerated, and have long in vivo half-life as well as the capacity to induce immune-mediated virus clearance. Their activities can be further enhanced by integration of their variable fragments (Fvs) into bispecific antibodies (bsAbs), affording simultaneous targeting of multiple epitopes to improve potency and breadth and/or to mitigate against viral escape by a single mutation. Here, we explore a bsAb strategy for generation of pan-ebolavirus and pan-filovirus immunotherapeutics. Filoviruses, including Ebola virus (EBOV), Sudan virus (SUDV), and Marburg virus (MARV), cause severe hemorrhagic fever. Although there are two FDA-approved mAb therapies for EBOV infection, these do not extend to other filoviruses. Here, we combine Fvs from broad ebolavirus mAbs to generate novel pan-ebolavirus bsAbs that are potently neutralizing, confer protection in mice, and are resistant to viral escape. Moreover, we combine Fvs from pan-ebolavirus mAbs with those of protective MARV mAbs to generate pan-filovirus protective bsAbs. These results provide guidelines for broad antiviral bsAb design and generate new immunotherapeutic candidates.
Copyright: © 2024 Wirchnianski et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
K.C. owns shares in Integrum Scientific LLC and Eitr Biologics, Inc, and has consulted for Axon Advisors, LLC. J.R.L. is a paid consultant for Celdara Medical, LLC.
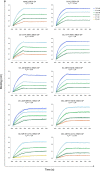
Figures




References
-
- Bwire G, Sartorius B, Guerin P, Tegegne MA, Okware SI, Talisuna AO. Sudan Ebola virus (SUDV) outbreak in Uganda, 2022: lessons learnt and future priorities for sub-Saharan Africa. BMC Med. 2023;21(1):144. Epub 2023/04/14. doi: 10.1186/s12916-023-02847-1 ; PubMed Central PMCID: PMC10099013. - DOI - PMC - PubMed
-
- Davey RT Jr., Dodd L, Proschan MA, Neaton J, Neuhaus Nordwall J, Koopmeiners JS, et al.. A Randomized, Controlled Trial of ZMapp for Ebola Virus Infection. The New England journal of medicine. 2016;375(15):1448–56. Epub 2016/10/13. doi: 10.1056/NEJMoa1604330 ; PubMed Central PMCID: PMC5086427. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

