Phase II trial of pembrolizumab and epacadostat in recurrent clear cell carcinoma of the ovary: An NRG oncology study GY016
- PMID: 38603953
- PMCID: PMC11265792
- DOI: 10.1016/j.ygyno.2024.03.027
Phase II trial of pembrolizumab and epacadostat in recurrent clear cell carcinoma of the ovary: An NRG oncology study GY016
Abstract
Introduction: Early reports of PD-1 inhibition in ovarian clear cell carcinomas (OCCC) demonstrate promising response. We evaluated the combination of pembrolizumab and IDO-1 inhibitor epacadostat in patients with recurrent OCCC.
Methods: This single arm, two-stage, phase 2 trial included those with measurable disease and 1-3 prior regimens. Patients received intravenous pembrolizumab 200 mg every 3 weeks and oral epacadostat 100 mg twice a day. Primary endpoint was overall response rate (ORR), secondary endpoints were toxicity, progression-free survival (PFS) and overall survival (OS). The study was powered to detect an absolute 25% increase in response (15% to 40%).
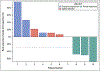
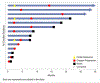
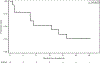
Results: Between September 28, 2018 and April 10, 2019, 14 patients enrolled at first stage. Rate of accrual was 2.3 patients per month. Median age was 65 years (44-89), 10 (71.4%) had ≥2 prior regimens. ORR was 21% (95% CI 5-51%) within 7 months of study entry with 3 partial responses, and 4 had stable disease (disease control rate 50%). Median PFS was 4.8 months (95% CI: 1.9-9.6), OS 18.9 months (95% CI: 1.9-NR). Most common grade ≥ 3 adverse events were electrolyte abnormalities and gastrointestinal pain, nausea, vomiting, bowel obstruction. In July 2019, the study reached the pre-specified criteria to re-open to second stage; however, the study closed prematurely in February 2021 due to insufficient drug supply.
Conclusions: Pembrolizumab and epacadostat demonstrated an ORR of 21% in this small cohort of recurrent OCCC. The rapid rate of accrual highlights the enthusiasm and need for therapeutic studies in patients with OCCC.
Keywords: Clear cell; IDO1; Ovarian; PD-1; Pembrolizumab.
Copyright © 2024 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest Dr. Lilian Gien received consulting fees from Merck – Advisor Board – October 2021. She also received Speaker Honorarium – January 2021 – from Merck. Dr. Danielle Enserro received funding from NCI for Cooperative Group/NCTN Grant Funding for all aspects of this trial including travel to Group meetings, trial design, statistical design and analysis, study monitoring, writing/editing of abstracts/manuscripts, etc. Dr. Matthew S. Block received grants or contracts from Merck – drug only contract for investigator-sponsored trial; Regeneron, Sorrento, Transgene, TILT Biotherapeutics, Alkermes, Bristol-Myers Squibb, Genentech, nFerence, Pharmacyclics and Viewpoint Molecular Therapeutics – institutional payment for clinical trial. He also has a patent filed for Dendritic Cell Based Vaccines Combined with Penbrolizumab for the Treatment of Advanced Ovarian Cancer – patent filed; author has waived rights to personal financial gain. He has participated on a Data Safety Monitoring Board or Advisory Board from TILT Biotherapeutics, Sorrento, and Viewpoint Molecular Therapeutics – no payment received. Dr. Linda Duska has multiple grants from sponsors for clinical trials. These grants go to her institution and not to her. These include, but are not limited to: (research funding (to institution) for investigator initiated trials for Merck, clinical trial grants (to institution) from Genentech/Roche, AbbVie/(GOG 3005), Acrivon, Advaxis, Aduro BioTech, Alkermes, Blueprint, Constellation, Eisai, GlaxoSmithKlein/Novartis, Immunogen, Inovio, Iovance, Karyopharm, KSQ Therapeutics, Lycera, Merck, Morab, MorphoTek, Naveris, Nurix, OncoQuest, Pfizer, Syndax, Tesaro, and Zentalis. She has Royalties or licenses (all up to date) with Wiley and ASCO (Editor of ASCO Connection). She received consulting fees from Regeneron, Aadi Bioscience and Merck for serving on Scientific Advisory Boards. She received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Advance Medical, CEA Group and Clinical Care Options (CME). She has participated on a Data Safety Monitoring Board or Advisory Board for Innovio DSMB (to institution) and Aegenus DSMB (to institution). She served as Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid as Secretary Treasurer for SGO (unpaid) and Editorial Board, British Journal of OB/GYN. Dr. Andrea E. Wahner-Hendrickson received Grants or contracts from TORL Therapeutics (funding to institution for clinical trial, OXCIA (advisory board – unpaid), Prolynx (funding to institution for clinical trial and Mayo Ovarian SPORE (P50 CA1363939). She participated on a Data Safety Monitoring Board or Advisory Board for OXCIA (unpaid). She also served in a Leadership or fiduciary role in other board, society, committee or advocacy group – MOCA (unpaid). Dr. Premal H. Thaker received grants to his institution from Merck and GlaxoSmithKline. She received consulting fees from Immunon. She also participated on a Data Safety Monitoring Board or Advisory Board with AstraZeneca, Clovis Oncology, GlaxoSmithKline, Seagen, Agenus, Immunon, Immuogen, Mersana, Novocure, R-Pharm, Zentalis, Aadi Pharmaceuticals, Merck, Caris Iovance and Verastem. She also has stock or stock options with Immunon. Dr. Floor Backes received grants or contracts from Merck, Eisai, ImmunoGen, Clovis, Beigene, Natera, Tempus and AstraZeneca (research grants paid to the institution). Royalties or licenses from UptoDate (personal fees). She also received consulting fees from Agenus, Merck, Clovis, Immunogen, Eisai, AstraZeneca, GlaxoSmithKline, Myriad, BioNTech and Daiichi Sankyo (Advisory boards – personal fees). She received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Clinical Educational Concepts, Clinical Care Options, Medscape/WebMD, Med Learngin, 13Health, CMR Institute, Global Learning Initiative/Prova, OncLive, Targeted Oncology and Research To Practice (CME lectures – personal fees). She received support for attending meetings and/or travel from GlaxoSmithKline. She participated on a Data Safety Monitoring Board – see consulting fees. She served in a Leadership or fiduciary role on other board, society, committee or advocacy group, paid or unpaid from Society of Gynecologic Oncology (Board member – unpaid), NRG Oncology Developmental Therapeutics Committee – Co-Chair and IGCS Education360 – Co-Chair. Dr. Carolyn Y. Muller received a grant to her institution from New Mexico Minority Underserved NCORP to support enrollment to all NCI NCTN trials. She has a contract to her institution to enroll to GOG Partners trials from GOG Partners Foundation (Segan, GSK, Mersana, Alkemes, Merck, Verastem, Immunogen, etc). She received contracts to her institution for enrollment to specific clinical trials from Linneus Therapeutics. She serves as Chair, Board of Directors of the New Mexico Cancer Research Alliance (unpaid position that manages an affiliate consortium to provide access to clinical trials across the states many health systems). Dr. Paul DiSilvestro serves on the NRG Oncology Board of Directors in a leadership or fiduciary role. Dr. David M. Gershenson's institution received a grant from Novartis. He has royalties or licenses from Elsevier and UpToDate. He received consulting fees to himself from Verastem. He received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Yale University and University of Washington. He serves in a Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid from International Consortium for Low Grade Serous Ovarian Cancer. He has stock in Bristol Myers Squibb, Johnson & Johnson and Proctor & Gamble. He has other financial or non-financial interests himself in Audi AB, Verastem AB, Springworks AB and Onconova AB. Dr. Kathleen N. Moore has grants/contracts from Clovis Oncology Pharmaceutical, Eli Lilly and Company, Genentech, GSK plc Pharmaceutical, Merck, PTC Therapeutics Pharmaceuticals, Verastem Oncology and Biotech. She received support for attending meetings and/or travel from Duality, GSK and Regeneron. She participates on a Data Safety Monitoring Board or Advisory Board for AstraZeneca, Aravive, Aadi Bioscience, Alkermes, Blueprint Medicines, Caris, Clovis Oncology Pharmaceutical, Duality, Eisai Pharmaceutical, EMD Serono Inc., Eli Lilly and Company, Genentech Biotechology, GSK plc Pharmaceutical, ImmunoGen Biotechnology, InxMed, I-MAB Biotech, Iovance, Jiangsu Hengrui Medicine Pharmaceutical, Merck, Mereo BioPharma Group, Mersana Therapeutics Inc., Myriad Genetics, Novartis Pharmaceuticals, Onconova Therapeutics Inc., OncXerna Therapeutics, Inc., Regeneron, VBL, and Verastem Oncology. She serves in a leadership or fiduciary role for GOG Partners and ASCO. Dr. Carol Aghajanian received Clinical Trial funding to her institution (MSK) from: Abbvie – MSKPI – GOG 3005; AstraZeneca – MSK PI, SOLO1/GOG 3004; National Coordinating Investigator and MSK PI, DO81RC00001; ENGOT-ov46; AGO-OVAR 23; GOG-3025; Clovis – MSK PI, ARIEL 2 &3; Genentech/Roche – MSK PI, GOG3015 (IMagyn050). She also participates on an Advisory Board for Blueprint Medicine – Advisory Board 6/30/21 (no consulting fee); Mrck – Global Cervical and Ovaian Cancer Virtual Advisory Board 7/10/23 (no consulting fee) and AstraZeneca – AZ Evolve dmc 4/26/23-ongoing, She also serves on the GOG Foundation, Board of Directors (unpaid, occasional travel cost reimbursement to attend meetings) and NRG Oncology Board of Directors (unpaid).
Figures

Similar articles
-
A randomized, open-label, phase 3 trial of pembrolizumab plus epacadostat versus sunitinib or pazopanib as first-line treatment for metastatic renal cell carcinoma (KEYNOTE-679/ECHO-302).BMC Cancer. 2024 Jul 25;23(Suppl 1):1253. doi: 10.1186/s12885-023-10971-7. BMC Cancer. 2024. PMID: 39054430 Free PMC article. Clinical Trial.
-
Rucaparib versus chemotherapy for treatment of relapsed ovarian cancer with deleterious BRCA1 or BRCA2 mutation (ARIEL4): final results of an international, open-label, randomised, phase 3 trial.Lancet Oncol. 2025 Feb;26(2):249-264. doi: 10.1016/S1470-2045(24)00674-0. Lancet Oncol. 2025. PMID: 39914419 Clinical Trial.
-
Lurbinectedin Plus Pembrolizumab in Relapsed SCLC: The Phase I/II LUPER Study.J Thorac Oncol. 2025 Jul;20(7):969-982. doi: 10.1016/j.jtho.2025.02.005. Epub 2025 Feb 10. J Thorac Oncol. 2025. PMID: 39938593 Clinical Trial.
-
Taxane monotherapy regimens for the treatment of recurrent epithelial ovarian cancer.Cochrane Database Syst Rev. 2022 Jul 12;7(7):CD008766. doi: 10.1002/14651858.CD008766.pub3. Cochrane Database Syst Rev. 2022. PMID: 35866378 Free PMC article.
-
Pembrolizumab monotherapy versus chemotherapy for treatment of advanced urothelial carcinoma with disease progression during or following platinum-containing chemotherapy. A Cochrane Rapid Review.Cochrane Database Syst Rev. 2018 Jul 23;7(7):CD012838. doi: 10.1002/14651858.CD012838.pub2. Cochrane Database Syst Rev. 2018. PMID: 30036453 Free PMC article.
Cited by
-
Genetic and therapeutic heterogeneity shape the baseline and longitudinal immune ecosystem of ovarian clear cell carcinoma.J Immunother Cancer. 2024 Nov 27;12(11):e010069. doi: 10.1136/jitc-2024-010069. J Immunother Cancer. 2024. PMID: 39608974 Free PMC article.
-
Infiltration of CD8+ cytotoxic T-cells and expression of PD-1 and PD-L1 in ovarian clear cell carcinoma.Sci Rep. 2025 Feb 8;15(1):4716. doi: 10.1038/s41598-025-89270-z. Sci Rep. 2025. PMID: 39922892 Free PMC article.
References
-
- Anglesio MS, Carey MS, Kobel M, MacKay H, Huntsman DG et al. Clear cell carcinoma of the ovary: a report from the first Ovarian Clear Cell Symposium. Gynecol Oncol 2011; 121:407–15. - PubMed
-
- Takano M, Sugiyama T, Yaegashi N, Sakuma M, Suzuki M, Saga Y et al. Low response rate of second-line chemotherapy for recurrent or refractory clear cell carcinoma of the ovary: a retrospective Japan Clear Cell Carcinoma study. Int J Gynecol Cancer 2008; 18:937–42. - PubMed
-
- Matulonis UA, Shapira-Frommer R, Santin A, Lisyanskaya AS, Pignata S, Vergote I et al. Antitumor activity and safety of pembrolizumab in patients with advanced recurrent ovarian cancer: interim results from the phase II KEYNOTE- 100 study, Ann Oncol 2019; 30:1080–7. - PubMed
-
- Hamanishi J, Mandai M, Ikeda T, Minami M, Kawaguchi A, Murayama T, et al. Safety and antitumor activity of anti-PD-1 antibody, nivolumab, in patients with platinum-resistant ovarian cancer. J Clin Oncol 2015; 33:4015–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

