Lipid exchange of apolipoprotein A-I amyloidogenic variants in reconstituted high-density lipoprotein with artificial membranes
- PMID: 38607188
- PMCID: PMC11010956
- DOI: 10.1002/pro.4987
Lipid exchange of apolipoprotein A-I amyloidogenic variants in reconstituted high-density lipoprotein with artificial membranes
Abstract
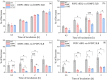
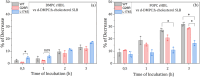
High-density lipoproteins (HDLs) are responsible for removing cholesterol from arterial walls, through a process known as reverse cholesterol transport. The main protein in HDL, apolipoprotein A-I (ApoA-I), is essential to this process, and changes in its sequence significantly alter HDL structure and functions. ApoA-I amyloidogenic variants, associated with a particular hereditary degenerative disease, are particularly effective at facilitating cholesterol removal, thus protecting carriers from cardiovascular disease. Thus, it is conceivable that reconstituted HDL (rHDL) formulations containing ApoA-I proteins with functional/structural features similar to those of amyloidogenic variants hold potential as a promising therapeutic approach. Here we explored the effect of protein cargo and lipid composition on the function of rHDL containing one of the ApoA-I amyloidogenic variants G26R or L174S by Fourier transformed infrared spectroscopy and neutron reflectometry. Moreover, small-angle x-ray scattering uncovered the structural and functional differences between rHDL particles, which could help to comprehend higher cholesterol efflux activity and apparent lower phospholipid (PL) affinity. Our findings indicate distinct trends in lipid exchange (removal vs. deposition) capacities of various rHDL particles, with the rHDL containing the ApoA-I amyloidogenic variants showing a markedly lower ability to remove lipids from artificial membranes compared to the rHDL containing the native protein. This effect strongly depends on the level of PL unsaturation and on the particles' ultrastructure. The study highlights the importance of the protein cargo, along with lipid composition, in shaping rHDL structure, contributing to our understanding of lipid-protein interactions and their behavior.
Keywords: amyloidogenic variants; apolipoprotein A‐I; high‐density lipoprotein; reconstituted HDL.
© 2024 The Authors. Protein Science published by Wiley Periodicals LLC on behalf of The Protein Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

 , CmSI;
, CmSI;  , d‐TBS;
, d‐TBS;  , h‐TBS) and after (
, h‐TBS) and after ( , CmSI;
, CmSI;  , d‐TBS;
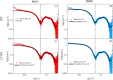
, d‐TBS;  , h‐TBS) incubation with 0.132 mg/mL rHDL containing the (a) POPC WT, (b) POPC L174S, (c) DMPC WT, and (d) DMPC L174S, measured at 37°C in d‐TBS, h‐TBS, and CmSi‐Tris. Left panel: SLD profiles of the SLB structures before (short dash) and after (solid line) incubation. Illustrations correspond to the SLB structure after incubation and rinse. ApoA‐I, apolipoprotein A‐I; d‐POPC, perdeuterated 1‐palmitoyl‐oleoyl‐d64‐3‐sn‐glycerophosphatidylcholine; DMPC, 1,2‐dimyristoyl‐d54‐3‐sn‐glycerophosphatidylcholine; h‐TBS, 50 mM Tris buffer, 150 mM NaCl, pH 7.4 in H2O; PLs, phospholipids; rHDL, reconstituted high‐density lipoprotein; SLB, supported lipid bilayer; SLD, scattering length density; WT, wild‐type.
, h‐TBS) incubation with 0.132 mg/mL rHDL containing the (a) POPC WT, (b) POPC L174S, (c) DMPC WT, and (d) DMPC L174S, measured at 37°C in d‐TBS, h‐TBS, and CmSi‐Tris. Left panel: SLD profiles of the SLB structures before (short dash) and after (solid line) incubation. Illustrations correspond to the SLB structure after incubation and rinse. ApoA‐I, apolipoprotein A‐I; d‐POPC, perdeuterated 1‐palmitoyl‐oleoyl‐d64‐3‐sn‐glycerophosphatidylcholine; DMPC, 1,2‐dimyristoyl‐d54‐3‐sn‐glycerophosphatidylcholine; h‐TBS, 50 mM Tris buffer, 150 mM NaCl, pH 7.4 in H2O; PLs, phospholipids; rHDL, reconstituted high‐density lipoprotein; SLB, supported lipid bilayer; SLD, scattering length density; WT, wild‐type.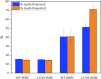
Similar articles
-
The structure of the apolipoprotein A-I monomer provides insights into its oligomerisation and lipid-binding mechanisms.J Mol Biol. 2025 Aug 13:169394. doi: 10.1016/j.jmb.2025.169394. Online ahead of print. J Mol Biol. 2025. PMID: 40816717
-
APOA2 increases cholesterol efflux capacity to plasma HDL by displacing the C-terminus of resident APOA1.J Lipid Res. 2024 Dec;65(12):100686. doi: 10.1016/j.jlr.2024.100686. Epub 2024 Oct 28. J Lipid Res. 2024. PMID: 39490930 Free PMC article.
-
The Role of APOA-I in Alzheimer's Disease: Bridging Peripheral Tissues and the Central Nervous System.Pharmaceuticals (Basel). 2025 May 25;18(6):790. doi: 10.3390/ph18060790. Pharmaceuticals (Basel). 2025. PMID: 40573187 Free PMC article. Review.
-
RETRACTED: The effects of Implanon on lipid metabolism in comparison with Norplant.Contraception. 1999 Nov;60(5):281-7. doi: 10.1016/s0010-7824(99)00099-2. Contraception. 1999. Retraction in: Contraception. 2004 Nov;70(5):433. doi: 10.1016/j.contraception.2004.07.004. Retraction in: Contraception. 2025 Aug;148:110947. doi: 10.1016/j.contraception.2025.110947. PMID: 10717780 Retracted. Clinical Trial.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

