Impact of Distinct Antiandrogen Exposures on the Plasma Metabolome in Feminizing Gender-affirming Hormone Therapy
- PMID: 38609170
- PMCID: PMC11479691
- DOI: 10.1210/clinem/dgae226
Impact of Distinct Antiandrogen Exposures on the Plasma Metabolome in Feminizing Gender-affirming Hormone Therapy
Abstract
Context: The plasma metabolome is a functional readout of metabolic activity and is associated with phenotypes exhibiting sexual dimorphism, such as cardiovascular disease. Sex hormones are thought to play a key role in driving sexual dimorphism.
Objective: Gender-affirming hormone therapy (GAHT) is a cornerstone of transgender care, but longitudinal changes in the plasma metabolome with feminizing GAHT have not been described.
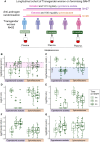
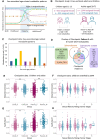
Methods: Blood samples were collected at baseline and after 3 and 6 months of GAHT from transgender women (n = 53). Participants were randomized to different anti-androgens, cyproterone acetate or spironolactone. Nuclear magnetic resonance-based metabolomics was used to measure 249 metabolic biomarkers in plasma. Additionally, we used metabolic biomarker data from an unrelated cohort of children and their parents (n = 3748) to identify sex- and age-related metabolite patterns.
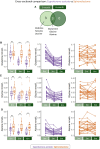
Results: We identified 43 metabolic biomarkers altered after 6 months in both anti-androgen groups, most belonging to the very low- or low-density lipoprotein subclasses, with all but 1 showing a decrease. We observed a cyproterone acetate-specific decrease in glutamine, glycine, and alanine levels. Notably, of the metabolic biomarkers exhibiting the most abundant "sex- and age-related" pattern (higher in assigned female children and lower in assigned female adults, relative to assigned males), 80% were significantly lowered after GAHT, reflecting a shift toward the adult female profile.
Conclusion: Our results suggest an anti-atherogenic signature in the plasma metabolome after the first 6 months of feminizing GAHT, with cyproterone acetate also reducing specific plasma amino acids. This study provides novel insight into the metabolic changes occurring across feminizing GAHT.
Keywords: GAHT; estradiol; metabolism; metabolome; sex hormones; transgender.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society.
Figures


Similar articles
-
Effects of gender affirming hormone treatment in transgender individuals - a retrospective cohort study.Endocrine. 2024 Jul;85(1):370-379. doi: 10.1007/s12020-024-03736-0. Epub 2024 Feb 22. Endocrine. 2024. PMID: 38386168
-
Anti-Androgenic Effects Comparison Between Cyproterone Acetate and Spironolactone in Transgender Women: A Randomized Controlled Trial.J Sex Med. 2021 Jul;18(7):1299-1307. doi: 10.1016/j.jsxm.2021.05.003. J Sex Med. 2021. PMID: 34274044 Clinical Trial.
-
Differential Effects of Cyproterone Acetate vs Spironolactone on Serum High-Density Lipoprotein and Prolactin Concentrations in the Hormonal Treatment of Transgender Women.J Sex Med. 2016 Nov;13(11):1765-1772. doi: 10.1016/j.jsxm.2016.09.012. Epub 2016 Sep 29. J Sex Med. 2016. PMID: 27693265
-
MANAGEMENT OF ENDOCRINE DISEASE: Optimal feminizing hormone treatment in transgender people.Eur J Endocrinol. 2021 Jun 28;185(2):R49-R63. doi: 10.1530/EJE-21-0059. Eur J Endocrinol. 2021. PMID: 34081614 Review.
-
REPRODUCTIVE HEALTH IN TRANS AND GENDER DIVERSE PATIENTS: Effects of feminizing gender-affirming hormone therapy on testicular function and reproductive capacity: review of data from clinical and experimental studies.Reproduction. 2025 Jan 9;169(2):e240046. doi: 10.1530/REP-24-0046. Print 2025 Feb 1. Reproduction. 2025. PMID: 39626030 Review.
Cited by
-
Epigenetic remodeling by sex hormone receptors and implications for gender affirming hormone therapy.Front Immunol. 2025 May 8;16:1501959. doi: 10.3389/fimmu.2025.1501959. eCollection 2025. Front Immunol. 2025. PMID: 40406098 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

