RUVBL1 ubiquitination by DTL promotes RUVBL1/2-β-catenin-mediated transcriptional regulation of NHEJ pathway and enhances radiation resistance in breast cancer
- PMID: 38609375
- PMCID: PMC11015013
- DOI: 10.1038/s41419-024-06651-4
RUVBL1 ubiquitination by DTL promotes RUVBL1/2-β-catenin-mediated transcriptional regulation of NHEJ pathway and enhances radiation resistance in breast cancer
Abstract
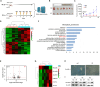
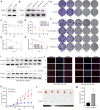
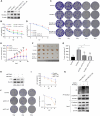
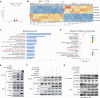
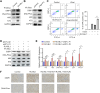
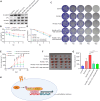
Radiotherapy effectiveness in breast cancer is limited by radioresistance. Nevertheless, the mechanisms behind radioresistance are not yet fully understood. RUVBL1 and RUVBL2, referred to as RUVBL1/2, are crucial AAA+ ATPases that act as co-chaperones and are connected to cancer. Our research revealed that RUVBL1, also known as pontin/TIP49, is excessively expressed in MMTV-PyMT mouse models undergoing radiotherapy, which is considered a murine spontaneous breast-tumor model. Our findings suggest that RUVBL1 enhances DNA damage repair and radioresistance in breast cancer cells both in vitro and in vivo. Mechanistically, we discovered that DTL, also known as CDT2 or DCAF2, which is a substrate adapter protein of CRL4, promotes the ubiquitination of RUVBL1 and facilitates its binding to RUVBL2 and transcription cofactor β-catenin. This interaction, in turn, attenuates its binding to acetyltransferase Tat-interacting protein 60 (TIP60), a comodulator of nuclear receptors. Subsequently, ubiquitinated RUVBL1 promotes the transcriptional regulation of RUVBL1/2-β-catenin on genes associated with the non-homologous end-joining (NHEJ) repair pathway. This process also attenuates TIP60-mediated H4K16 acetylation and the homologous recombination (HR) repair process. Expanding upon the prior study's discoveries, we exhibited that the ubiquitination of RUVBL1 by DTL advances the interosculation of RUVBL1/2-β-catenin. And, it then regulates the transcription of NHEJ repair pathway protein. Resulting in an elevated resistance of breast cancer cells to radiation therapy. From the aforementioned, it is evident that targeting DTL-RUVBL1/2-β-catenin provides a potential radiosensitization approach when treating breast cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
CircMYO10 promotes osteosarcoma progression by regulating miR-370-3p/RUVBL1 axis to enhance the transcriptional activity of β-catenin/LEF1 complex via effects on chromatin remodeling.Mol Cancer. 2019 Oct 29;18(1):150. doi: 10.1186/s12943-019-1076-1. Mol Cancer. 2019. PMID: 31665067 Free PMC article.
-
PRMT5-Dependent Methylation of the TIP60 Coactivator RUVBL1 Is a Key Regulator of Homologous Recombination.Mol Cell. 2017 Mar 2;65(5):900-916.e7. doi: 10.1016/j.molcel.2017.01.019. Epub 2017 Feb 23. Mol Cell. 2017. PMID: 28238654 Free PMC article.
-
Regulation of the β‑catenin/LEF‑1 pathway by the siRNA knockdown of RUVBL1 expression inhibits breast cancer cell proliferation, migration and invasion.Oncol Rep. 2025 Feb;53(2):22. doi: 10.3892/or.2024.8855. Epub 2024 Dec 13. Oncol Rep. 2025. PMID: 39670302 Free PMC article.
-
RUVBL1-RUVBL2 AAA-ATPase: a versatile scaffold for multiple complexes and functions.Curr Opin Struct Biol. 2021 Apr;67:78-85. doi: 10.1016/j.sbi.2020.08.010. Epub 2020 Oct 28. Curr Opin Struct Biol. 2021. PMID: 33129013 Review.
-
The Role of Pontin and Reptin in Cellular Physiology and Cancer Etiology.Front Mol Biosci. 2017 Aug 24;4:58. doi: 10.3389/fmolb.2017.00058. eCollection 2017. Front Mol Biosci. 2017. PMID: 28884116 Free PMC article. Review.
Cited by
-
Rewired glycolysis by DTL accelerates oncometabolite L-lactate generation to promote breast cancer progression.Front Oncol. 2025 May 5;15:1583752. doi: 10.3389/fonc.2025.1583752. eCollection 2025. Front Oncol. 2025. PMID: 40391156 Free PMC article.
-
Overview of Wnt/β-Catenin Pathway and DNA Damage/Repair in Cancer.Biology (Basel). 2025 Feb 11;14(2):185. doi: 10.3390/biology14020185. Biology (Basel). 2025. PMID: 40001953 Free PMC article. Review.
-
Evaluating the Cellular Roles of the Lysine Acetyltransferase Tip60 in Cancer: A Multi-Action Molecular Target for Precision Oncology.Cancers (Basel). 2024 Jul 27;16(15):2677. doi: 10.3390/cancers16152677. Cancers (Basel). 2024. PMID: 39123405 Free PMC article. Review.
-
Establishment and transcriptomic characteristics of radio-resistant meningioma cell lines.J Neurooncol. 2025 May;173(1):105-119. doi: 10.1007/s11060-025-04966-6. Epub 2025 Feb 28. J Neurooncol. 2025. PMID: 40019713
References
-
- Sjostrom M, Lundstedt D, Hartman L, Holmberg E, Killander F, Kovacs A, et al. Response to radiotherapy after breast-conserving surgery in different breast cancer subtypes in the Swedish Breast Cancer Group 91 Radiotherapy Randomized Clinical Trial. J Clin Oncol. 2017;35:3222–9. doi: 10.1200/JCO.2017.72.7263. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

