Rel Family Transcription Factor NFAT5 Upregulates COX2 via HIF-1α Activity in Ishikawa and HEC1a Cells
- PMID: 38612478
- PMCID: PMC11012216
- DOI: 10.3390/ijms25073666
Rel Family Transcription Factor NFAT5 Upregulates COX2 via HIF-1α Activity in Ishikawa and HEC1a Cells
Abstract
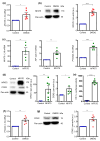
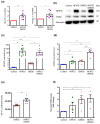
Nuclear factor of activated T cells 5 (NFAT5) and cyclooxygenase 2 (COX2; PTGS2) both participate in diverse pathologies including cancer progression. However, the biological role of the NFAT5-COX2 signaling pathway in human endometrial cancer has remained elusive. The present study explored whether NFAT5 is expressed in endometrial tumors and if NFAT5 participates in cancer progression. To gain insights into the underlying mechanisms, NFAT5 protein abundance in endometrial cancer tissue was visualized by immunohistochemistry and endometrial cancer cells (Ishikawa and HEC1a) were transfected with NFAT5 or with an empty plasmid. As a result, NFAT5 expression is more abundant in high-grade than in low-grade endometrial cancer tissue. RNA sequencing analysis of NFAT5 overexpression in Ishikawa cells upregulated 37 genes and downregulated 20 genes. Genes affected included cyclooxygenase 2 and hypoxia inducible factor 1α (HIF1A). NFAT5 transfection and/or treatment with HIF-1α stabilizer exerted a strong stimulating effect on HIF-1α promoter activity as well as COX2 expression level and prostaglandin E2 receptor (PGE2) levels. Our findings suggest that activation of NFAT5-HIF-1α-COX2 axis could promote endometrial cancer progression.
Keywords: cyclooxygenase 2; endometrial cancer; hypoxia inducible factor 1α.
Conflict of interest statement
The authors declare that they have no conflicts of interests.
Figures



Similar articles
-
Increased NFAT5 expression stimulates transcription of Hsp70 in preeclamptic placentas.Placenta. 2014 Feb;35(2):109-16. doi: 10.1016/j.placenta.2013.12.005. Epub 2013 Dec 21. Placenta. 2014. PMID: 24398013
-
JAK1 inactivation promotes proliferation and migration of endometrial cancer cells via upregulating the hypoxia-inducible factor signaling pathway.Cell Commun Signal. 2022 Nov 14;20(1):177. doi: 10.1186/s12964-022-00990-5. Cell Commun Signal. 2022. PMID: 36376931 Free PMC article.
-
Prostaglandin E2 induces hypoxia-inducible factor-1alpha stabilization and nuclear localization in a human prostate cancer cell line.J Biol Chem. 2002 Dec 20;277(51):50081-6. doi: 10.1074/jbc.M201095200. Epub 2002 Oct 24. J Biol Chem. 2002. PMID: 12401798
-
Hypoxia-induced factor-1α in endometrial carcinoma: a mini-review of current evidence.Histol Histopathol. 2012 Oct;27(10):1247-53. doi: 10.14670/HH-27.1247. Histol Histopathol. 2012. PMID: 22936443 Review.
-
Prognostic and Clinicopathological Significance of Hypoxia-Inducible Factor-1α in Endometrial Cancer: A Meta-Analysis.Front Oncol. 2020 Nov 11;10:587420. doi: 10.3389/fonc.2020.587420. eCollection 2020. Front Oncol. 2020. PMID: 33304847 Free PMC article.
Cited by
-
NFAT5: a stress-related transcription factor with multiple functions in health and disease.Cell Stress. 2025 May 22;9:16-48. doi: 10.15698/cst2025.05.304. eCollection 2025. Cell Stress. 2025. PMID: 40421201 Free PMC article. Review.
-
Nuclear Factor of Activated T Cells (NFAT) Proteins as Targeted Molecules in Diseases: A Narrative Review.Cureus. 2024 Dec 16;16(12):e75844. doi: 10.7759/cureus.75844. eCollection 2024 Dec. Cureus. 2024. PMID: 39822413 Free PMC article. Review.
-
Placental growth factor mediates pathological uterine angiogenesis by activating the NFAT5-SGK1 signaling axis in the endometrium: implications for preeclampsia development.Biol Res. 2024 Aug 17;57(1):55. doi: 10.1186/s40659-024-00526-w. Biol Res. 2024. PMID: 39152497 Free PMC article.
-
During high salt treatment myeloid p38α/MAPK fosters osteoclast activity and inflammatory macrophage responses promoting orthodontic tooth movement.Front Immunol. 2025 Apr 15;16:1571268. doi: 10.3389/fimmu.2025.1571268. eCollection 2025. Front Immunol. 2025. PMID: 40303394 Free PMC article.
References
-
- Leone Roberti Maggiore U., Spanò Bascio L., Alboni C., Chiarello G., Savelli L., Bogani G., Martinelli F., Chiappa V., Ditto A., Raspagliesi F. Sentinel lymph node biopsy in endometrial cancer: When, how and in which patients. Eur. J. Surg. Oncol. 2024;50:107956. doi: 10.1016/j.ejso.2024.107956. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

