Kaempferol as an Alternative Cryosupplement for Bovine Spermatozoa: Cytoprotective and Membrane-Stabilizing Effects
- PMID: 38612937
- PMCID: PMC11012659
- DOI: 10.3390/ijms25074129
Kaempferol as an Alternative Cryosupplement for Bovine Spermatozoa: Cytoprotective and Membrane-Stabilizing Effects
Abstract
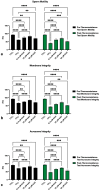
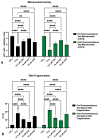
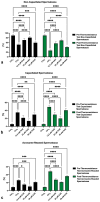
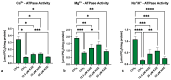
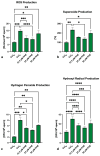
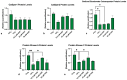
Kaempferol (KAE) is a natural flavonoid with powerful reactive oxygen species (ROS) scavenging properties and beneficial effects on ex vivo sperm functionality. In this paper, we studied the ability of KAE to prevent or ameliorate structural, functional or oxidative damage to frozen-thawed bovine spermatozoa. The analysis focused on conventional sperm quality characteristics prior to or following thermoresistance tests, namely the oxidative profile of semen alongside sperm capacitation patterns, and the levels of key proteins involved in capacitation signaling. Semen samples obtained from 30 stud bulls were frozen in the presence of 12.5, 25 or 50 μM KAE and compared to native ejaculates (negative control-CtrlN) as well as semen samples cryopreserved in the absence of KAE (positive control-CtrlC). A significant post-thermoresistance test maintenance of the sperm motility (p < 0.001), membrane (p < 0.001) and acrosome integrity (p < 0.001), mitochondrial activity (p < 0.001) and DNA integrity (p < 0.001) was observed following supplementation with all KAE doses in comparison to CtrlC. Experimental groups supplemented with all KAE doses presented a significantly lower proportion of prematurely capacitated spermatozoa (p < 0.001) when compared with CtrlC. A significant decrease in the levels of the superoxide radical was recorded following administration of 12.5 (p < 0.05) and 25 μM KAE (p < 0.01). At the same time, supplementation with 25 μM KAE in the cryopreservation medium led to a significant stabilization of the activity of Mg2+-ATPase (p < 0.05) and Na+/K+-ATPase (p < 0.0001) in comparison to CtrlC. Western blot analysis revealed that supplementation with 25 μM KAE in the cryopreservation medium prevented the loss of the protein kinase A (PKA) and protein kinase C (PKC), which are intricately involved in the process of sperm activation. In conclusion, we may speculate that KAE is particularly efficient in the protection of sperm metabolism during the cryopreservation process through its ability to promote energy synthesis while quenching excessive ROS and to protect enzymes involved in the process of sperm capacitation and hyperactivation. These properties may provide supplementary protection to spermatozoa undergoing the freeze-thaw process.
Keywords: Western blot; bulls; cryocapacitation; kaempferol; reactive oxygen species; sperm cryopreservation.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Cryoprotective Potential of Theobromine in the Improvement of the Post-Thaw Quality of Bovine Spermatozoa.Cells. 2024 Oct 16;13(20):1710. doi: 10.3390/cells13201710. Cells. 2024. PMID: 39451229 Free PMC article.
-
Epicatechin Prevents Cryocapacitation of Bovine Spermatozoa through Antioxidant Activity and Stabilization of Transmembrane Ion Channels.Int J Mol Sci. 2023 Jan 28;24(3):2510. doi: 10.3390/ijms24032510. Int J Mol Sci. 2023. PMID: 36768832 Free PMC article.
-
ROS-induced oxidative stress is a major contributor to sperm cryoinjury.Hum Reprod. 2024 Feb 1;39(2):310-325. doi: 10.1093/humrep/dead250. Hum Reprod. 2024. PMID: 38011909
-
Strategies to Minimize Various Stress-Related Freeze-Thaw Damages During Conventional Cryopreservation of Mammalian Spermatozoa.Biopreserv Biobank. 2019 Dec;17(6):603-612. doi: 10.1089/bio.2019.0037. Epub 2019 Aug 20. Biopreserv Biobank. 2019. PMID: 31429586 Review.
-
Potential Use of Tannin Extracts as Additives in Semen Destined for Cryopreservation: A Review.Animals (Basel). 2022 Apr 28;12(9):1130. doi: 10.3390/ani12091130. Animals (Basel). 2022. PMID: 35565556 Free PMC article. Review.
Cited by
-
Cryoprotective Potential of Theobromine in the Improvement of the Post-Thaw Quality of Bovine Spermatozoa.Cells. 2024 Oct 16;13(20):1710. doi: 10.3390/cells13201710. Cells. 2024. PMID: 39451229 Free PMC article.
-
Immunomodulatory Potential of Kaempferol Isolated from Peronema canescens Jack. Leaves Through Inhibition of IL-6 Expression.Int J Mol Sci. 2025 Mar 27;26(7):3068. doi: 10.3390/ijms26073068. Int J Mol Sci. 2025. PMID: 40243778 Free PMC article.
-
Sperm Membrane: Molecular Implications and Strategies for Cryopreservation in Productive Species.Animals (Basel). 2025 Jun 19;15(12):1808. doi: 10.3390/ani15121808. Animals (Basel). 2025. PMID: 40564358 Free PMC article. Review.
References
-
- Peris-Frau P., Soler A.J., Iniesta-Cuerda M., Martín-Maestro A., Sánchez-Ajofrín I., Medina-Chávez D.A., Fernández-Santos M.R., García-Álvarez O., Maroto-Morales A., Montoro V., et al. Sperm Cryodamage in Ruminants: Understanding the Molecular Changes Induced by the Cryopreservation Process to Optimize Sperm Quality. Int. J. Mol. Sci. 2020;21:2781. doi: 10.3390/ijms21082781. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

