The key role of altered tubule cell lipid metabolism in kidney disease development
- PMID: 38614389
- PMCID: PMC11193624
- DOI: 10.1016/j.kint.2024.02.025
The key role of altered tubule cell lipid metabolism in kidney disease development
Abstract
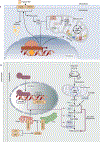
Kidney epithelial cells have very high energy requirements, which are largely met by fatty acid oxidation. Complex changes in lipid metabolism are observed in patients with kidney disease. Defects in fatty acid oxidation and increased lipid uptake, especially in the context of hyperlipidemia and proteinuria, contribute to this excess lipid build-up and exacerbate kidney disease development. Recent studies have also highlighted the role of increased de novo lipogenesis in kidney fibrosis. The defect in fatty acid oxidation causes energy starvation. Increased lipid uptake, synthesis, and lower fatty acid oxidation can cause toxic lipid build-up, reactive oxygen species generation, and mitochondrial damage. A better understanding of these metabolic processes may open new treatment avenues for kidney diseases by targeting lipid metabolism.
Keywords: acute kidney injury; chronic kidney disease; de novo lipogenesis; fatty acid oxidation; lipid; metabolism.
Copyright © 2024 International Society of Nephrology. All rights reserved.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

