Additively manufactured bioceramic scaffolds based on triply periodic minimal surfaces for bone regeneration
- PMID: 38617462
- PMCID: PMC11010742
- DOI: 10.1177/20417314241244997
Additively manufactured bioceramic scaffolds based on triply periodic minimal surfaces for bone regeneration
Abstract
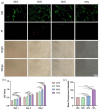
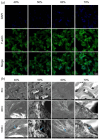
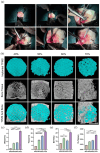
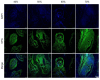
The study focused on the effects of a triply periodic minimal surface (TPMS) scaffolds, varying in porosity, on the repair of mandibular defects in New Zealand white rabbits. Four TPMS configurations (40%, 50%, 60%, and 70% porosity) were fabricated with β-tricalcium phosphate bioceramic via additive manufacturing. Scaffold properties were assessed through scanning electron microscopy and mechanical testing. For proliferation and adhesion assays, mouse bone marrow stem cells (BMSCs) were cultured on these scaffolds. In vivo, the scaffolds were implanted into rabbit mandibular defects for 2 months. Histological staining evaluated osteogenic potential. Moreover, RNA-sequencing analysis and RT-qPCR revealed the significant involvement of angiogenesis-related factors and Hippo signaling pathway in influencing BMSCs behavior. Notably, the 70% porosity TPMS scaffold exhibited optimal compressive strength, superior cell proliferation, adhesion, and significantly enhanced osteogenesis and angiogenesis. These findings underscore the substantial potential of 70% porosity TPMS scaffolds in effectively promoting bone regeneration within mandibular defects.
Keywords: 3D printer/additive manufacture; TPMS bone scaffold; bone regeneration; mandibular defect; osteogenesis.
© The Author(s) 2024.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures



Similar articles
-
The design of strut/TPMS-based pore geometries in bioceramic scaffolds guiding osteogenesis and angiogenesis in bone regeneration.Mater Today Bio. 2023 May 18;20:100667. doi: 10.1016/j.mtbio.2023.100667. eCollection 2023 Jun. Mater Today Bio. 2023. PMID: 37273795 Free PMC article.
-
Custom-made macroporous bioceramic implants based on triply-periodic minimal surfaces for bone defects in load-bearing sites.Acta Biomater. 2020 Jun;109:254-266. doi: 10.1016/j.actbio.2020.03.016. Epub 2020 Mar 17. Acta Biomater. 2020. PMID: 32194263
-
Gaussian curvature-driven direction of cell fate toward osteogenesis with triply periodic minimal surface scaffolds.Proc Natl Acad Sci U S A. 2022 Oct 11;119(41):e2206684119. doi: 10.1073/pnas.2206684119. Epub 2022 Oct 3. Proc Natl Acad Sci U S A. 2022. PMID: 36191194 Free PMC article.
-
Additively manufactured porous scaffolds by design for treatment of bone defects.Front Bioeng Biotechnol. 2024 Jan 19;11:1252636. doi: 10.3389/fbioe.2023.1252636. eCollection 2023. Front Bioeng Biotechnol. 2024. PMID: 38312510 Free PMC article. Review.
-
Biomimetic scaffolds using triply periodic minimal surface-based porous structures for biomedical applications.SLAS Technol. 2023 Jun;28(3):165-182. doi: 10.1016/j.slast.2023.04.004. Epub 2023 Apr 29. SLAS Technol. 2023. PMID: 37127136 Review.
Cited by
-
Layered Gradient Rhombic Dodecahedron Composite Structures for Biomimetic Bone Fabricated via Selective Laser Melting.Micromachines (Basel). 2025 May 31;16(6):673. doi: 10.3390/mi16060673. Micromachines (Basel). 2025. PMID: 40572391 Free PMC article.
-
Bi-phasic integrated silk fibroin/polycaprolactone scaffolds for osteochondral regeneration inspired by the native joint tissue and interface.Mater Today Bio. 2025 Apr 8;32:101737. doi: 10.1016/j.mtbio.2025.101737. eCollection 2025 Jun. Mater Today Bio. 2025. PMID: 40275950 Free PMC article.
-
Novel 3D printed TPMS scaffolds: microstructure, characteristics and applications in bone regeneration.J Tissue Eng. 2024 Jul 26;15:20417314241263689. doi: 10.1177/20417314241263689. eCollection 2024 Jan-Dec. J Tissue Eng. 2024. PMID: 39071895 Free PMC article. Review.
-
Design of 3D printing osteotomy block for foot based on triply periodic minimal surface.Sci Rep. 2024 Jul 9;14(1):15851. doi: 10.1038/s41598-024-65318-4. Sci Rep. 2024. PMID: 38982110 Free PMC article.
References
-
- Ferguson BM, Entezari A, Fang J, et al.. Optimal placement of fixation system for scaffold-based mandibular reconstruction. J o Mech Behav Biomed Mater 2022; 126: 104855. - PubMed
-
- Brown JS, Khan A, Wareing S, et al.. A new classification of mandibular fractures. Int J Oral Maxillofac Surg 2022; 51(1): 78–90. - PubMed
LinkOut - more resources
Full Text Sources

