BRAFV600E-mutant metastatic NSCLC: disease overview and treatment landscape
- PMID: 38627602
- PMCID: PMC11021522
- DOI: 10.1038/s41698-024-00552-7
BRAFV600E-mutant metastatic NSCLC: disease overview and treatment landscape
Erratum in
-
Author Correction: BRAFV600E-mutant metastatic NSCLC: disease overview and treatment landscape.NPJ Precis Oncol. 2024 Jul 15;8(1):142. doi: 10.1038/s41698-024-00633-7. NPJ Precis Oncol. 2024. PMID: 39009670 Free PMC article. No abstract available.
Abstract
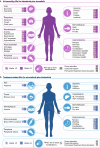
In this review, we cover the current understanding of BRAF mutations and associated clinical characteristics in patients with metastatic NSCLC, approved and emerging treatment options, BRAF sequencing approaches, and unmet needs. The BRAFV600E mutation confers constitutive activity of the MAPK pathway, leading to enhanced growth, proliferation, and survival of tumor cells. Testing for BRAF mutations enables patients to be treated with therapies that directly target BRAFV600E and the MAPK pathway, but BRAF testing lags behind other oncogene testing in metastatic NSCLC. Additional therapies targeting BRAFV600E mutations provide options for patients with metastatic NSCLC. Emerging therapies and combinations under investigation could potentially overcome issues of resistance and target non-V600E mutations. Therefore, because targeted therapies with enhanced efficacy are on the horizon, being able to identify BRAF mutations in metastatic NSCLC may become even more important.
© 2024. The Author(s).
Conflict of interest statement
D.P. reports receiving honoraria from Prime Oncology and PeerVoice; and has had a consulting or advisory role for AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Novartis, Roche, Pfizer, Merck Sharp & Dohme Oncology, Celgene, MedImmune, BeiGene, Samsung, AbbVie, Janssen, Seagen, Takeda, ArriVent, Mirati, Pierre Fabre, and Daiichi Sankyo/AstraZeneca; and has received research funding (institutional financial interest, clinical trials research as principal or co-investigator) from AstraZeneca/MedImmune, Bristol Myers Squibb, Boehringer Ingelheim, Lilly, Merck, Novartis, Pfizer, Roche, Sanofi/Aventis, Taiho Pharmaceutical, Daiichi Sankyo, AbbVie, Janssen, ArriVent, Mirati, Pierre Fabre, Seagen, and Takeda. R.S. reports receiving honoraria from AstraZeneca, GameOn!, Illumina, OncLive, and Targeted Oncology; and has had a consulting or advisory role for Amgen, AstraZeneca, EMD Serono, Daiichi Sankyo, G1 Therapeutics, GE HealthCare, Gilead, GlaxoSmithKline, Janssen Oncology, Regeneron, Sanofi Aventis; and has received research funding from Bristol Myers Squibb, Merck, and AstraZeneca. M.N. reports receiving research funding to institution from Mirati, Novartis, Checkmate, Alaunos, AstraZeneca, Pfizer, Genentech, Navire; a consultant or advisory role for Mirati, Merck/MSD, Novartis, Genentech, Sanofi; and other support from Ziopharm Oncology, ApotheCom, Ashfield Healthcare. A.V. reports no conflicts of interest. E.S. reports receiving honoraria from AstraZeneca, Daiichi Sankyo/AstraZeneca, Merck KGaA, and Boehringer Ingelheim; and has had a consulting or advisory role at Lilly, AstraZeneca, Boehringer Ingelheim, Roche/Genentech, Bristol Myers Squibb, Merck KGaA, MSD Oncology, Takeda, Bayer, Novartis, Daiichi Sankyo, and Seagen; and has received research funding from Boehringer Ingelheim, Bayer, Roche/Genentech, AstraZeneca, and Bristol Myers Squibb.
Figures


References
-
- Abdayem PA, Planchard D. Ongoing progress in BRAF-mutated non-small cell lung cancer. Clin. Adv. Hematol. Oncol. 2022;20:662–672. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

