Multiple adverse outcomes associated with antipsychotic use in people with dementia: population based matched cohort study
- PMID: 38631737
- PMCID: PMC11022137
- DOI: 10.1136/bmj-2023-076268
Multiple adverse outcomes associated with antipsychotic use in people with dementia: population based matched cohort study
Abstract
Objective: To investigate risks of multiple adverse outcomes associated with use of antipsychotics in people with dementia.
Design: Population based matched cohort study.
Setting: Linked primary care, hospital and mortality data from Clinical Practice Research Datalink (CPRD), England.
Population: Adults (≥50 years) with a diagnosis of dementia between 1 January 1998 and 31 May 2018 (n=173 910, 63.0% women). Each new antipsychotic user (n=35 339, 62.5% women) was matched with up to 15 non-users using incidence density sampling.
Main outcome measures: The main outcomes were stroke, venous thromboembolism, myocardial infarction, heart failure, ventricular arrhythmia, fracture, pneumonia, and acute kidney injury, stratified by periods of antipsychotic use, with absolute risks calculated using cumulative incidence in antipsychotic users versus matched comparators. An unrelated (negative control) outcome of appendicitis and cholecystitis combined was also investigated to detect potential unmeasured confounding.
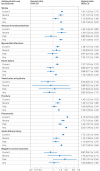
Results: Compared with non-use, any antipsychotic use was associated with increased risks of all outcomes, except ventricular arrhythmia. Current use (90 days after a prescription) was associated with elevated risks of pneumonia (hazard ratio 2.19, 95% confidence interval (CI) 2.10 to 2.28), acute kidney injury (1.72, 1.61 to 1.84), venous thromboembolism (1.62, 1.46 to 1.80), stroke (1.61, 1.52 to 1.71), fracture (1.43, 1.35 to 1.52), myocardial infarction (1.28, 1.15 to 1.42), and heart failure (1.27, 1.18 to 1.37). No increased risks were observed for the negative control outcome (appendicitis and cholecystitis). In the 90 days after drug initiation, the cumulative incidence of pneumonia among antipsychotic users was 4.48% (4.26% to 4.71%) versus 1.49% (1.45% to 1.53%) in the matched cohort of non-users (difference 2.99%, 95% CI 2.77% to 3.22%).
Conclusions: Antipsychotic use compared with non-use in adults with dementia was associated with increased risks of stroke, venous thromboembolism, myocardial infarction, heart failure, fracture, pneumonia, and acute kidney injury, but not ventricular arrhythmia. The range of adverse outcomes was wider than previously highlighted in regulatory alerts, with the highest risks soon after initiation of treatment.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: BG reports research grants from the National Institute for Health and Care Research (NIHR). DRM was awarded a Wellcome Trust Clinical Research Development Fellowship (214588/Z/18/Z). AS reports a research grant from the NIHR. RAE reports research grants from the NIHR and NHS England, and travel costs to attend a roundtable dinner discussion on medication errors, House of Commons, Westminster, on 29 March 2022. TvS reports research grants from the NIHR. AJA is national clinical director for prescribing for NHS England and reports research grants from the NIHR. DMA reports research grants from the NIHR, AbbVie, Almirall, Celgene, Eli Lilly, Janssen, Novartis, UCB, and the Leo Foundation. All other authors declare no support from any organisation for the submitted work (except those listed in the funding section); no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
Figures
References
-
- Stocks SJ, Kontopantelis E, Webb RT, Avery AJ, Burns A, Ashcroft DM. Antipsychotic prescribing to patients diagnosed with dementia without a diagnosis of psychosis in the context of national guidance and drug safety warnings: longitudinal study in UK general practice. Drug Saf 2017;40:679-92. 10.1007/s40264-017-0538-x - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
