The fasciola cinereum of the hippocampal tail as an interventional target in epilepsy
- PMID: 38632391
- PMCID: PMC11108783
- DOI: 10.1038/s41591-024-02924-9
The fasciola cinereum of the hippocampal tail as an interventional target in epilepsy
Abstract
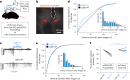
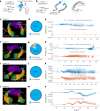
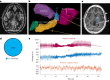
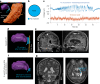
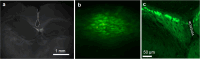
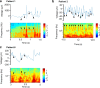
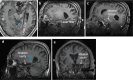
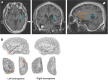
Targeted tissue ablation involving the anterior hippocampus is the standard of care for patients with drug-resistant mesial temporal lobe epilepsy. However, a substantial proportion continues to suffer from seizures even after surgery. We identified the fasciola cinereum (FC) neurons of the posterior hippocampal tail as an important seizure node in both mice and humans with epilepsy. Genetically defined FC neurons were highly active during spontaneous seizures in epileptic mice, and closed-loop optogenetic inhibition of these neurons potently reduced seizure duration. Furthermore, we specifically targeted and found the prominent involvement of FC during seizures in a cohort of six patients with epilepsy. In particular, targeted lesioning of the FC in a patient reduced the seizure burden present after ablation of anterior mesial temporal structures. Thus, the FC may be a promising interventional target in epilepsy.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
MeSH terms
Grants and funding
- R25NS065741/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R00 NS126725/NS/NINDS NIH HHS/United States
- P30 AG066515/AG/NIA NIH HHS/United States
- P30AG066515/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- 225914/Z/22/Z/Wellcome Trust (Wellcome)
- K99 NS126725/NS/NINDS NIH HHS/United States
- R25 NS065741/NS/NINDS NIH HHS/United States
- K99NS121399/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R01 NS121106/NS/NINDS NIH HHS/United States
- K99NS126725/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- R00 NS121399/NS/NINDS NIH HHS/United States
- K99 NS121399/NS/NINDS NIH HHS/United States
- R01NS121106/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- WT_/Wellcome Trust/United Kingdom
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

