Utility of bronchoscopically obtained frozen cytology pellets for next-generation sequencing
- PMID: 38632507
- PMCID: PMC11022476
- DOI: 10.1186/s12885-024-12250-5
Utility of bronchoscopically obtained frozen cytology pellets for next-generation sequencing
Abstract
Background: Next-generation sequencing (NGS) is essential for lung cancer treatment. It is important to collect sufficient tissue specimens, but sometimes we cannot obtain large enough samples for NGS analysis. We investigated the yield of NGS analysis by frozen cytology pellets using an Oncomine Comprehensive Assay or Oncomine Precision Assay.
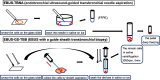
Methods: We retrospectively enrolled patients with lung cancer who underwent bronchoscopy at Kobe University Hospital and were enrolled in the Lung Cancer Genomic Screening Project for Individualized Medicine. We investigated the amount of extracted DNA and RNA and determined the NGS success rates. We also compared the amount of DNA and RNA by bronchoscopy methods. To create the frozen cytology pellets, we first effectively collected the cells and then quickly centrifuged and cryopreserved them.
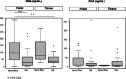
Results: A total of 132 patients were enrolled in this study between May 2016 and December 2022; of them, 75 were subjected to frozen cytology pellet examinations and 57 were subjected to frozen tissue examinations. The amount of DNA and RNA obtained by frozen cytology pellets was nearly equivalent to frozen tissues. Frozen cytology pellets collected by endobronchial ultrasound-guided transbronchial needle aspiration yielded significantly more DNA than those collected by transbronchial biopsy methods. (P < 0.01) In RNA content, cytology pellets were not inferior to frozen tissue. The success rate of NGS analysis with frozen cytology pellet specimens was comparable to the success rate of NGS analysis with frozen tissue specimens.
Conclusions: Our study showed that frozen cytology pellets may have equivalent diagnostic value to frozen tissue for NGS analyses. Bronchial cytology specimens are usually used only for cytology, but NGS analysis is possible if enough cells are collected to create pellet specimens. In particular, the frozen cytology pellets obtained by endobronchial ultrasound-guided transbronchial needle aspiration yielded sufficient amounts of DNA.
Trial registration: This was registered with the University Medical Hospital Information Network in Japan (UMINCTR registration no. UMIN000052050).
Keywords: Cytology; Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA); Lung cancer; Next-generation sequencing (NGS); Pellet specimen.
© 2024. The Author(s).
Conflict of interest statement
MT reports grants from AstraZeneca KK, Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan Co., Ltd., honoraria for lecture from Eli Lilly Japan Co., Ltd., Ono Pharmaceutical Co., Ltd., Bristol-Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., AstraZeneca KK, MSD KK, Novartis Pharmaceuticals K.K, Takeda Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co.,Ltd., Daiichi Sankyo, Pfizer Japan Inc., and Janssen Pharmaceutical K.K. SM reports grants from Chugai Pharma, Janssen Pharmaceutical, MSD, Novartis, honoraria for lecture from Eli Lilly, Merck Biopharma, Chugai Pharma, Novartis pharma, Guardant Health, and AstraZeneca. KG reports all support for the present manuscript from Thermo Fisher Scientific K.K., grants from Amgen Inc., Amgen K.K., Amgen Astellas BioPharma K.K., AstraZeneca K.K., Bayer Yakuhin, Ltd., Boehringer Ingelheim Japan, Inc., Bristol-Myers Squibb K.K., Chugai Pharmaceutical Co., Ltd., Craif Inc., DAIICHI SANKYO Co., Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., Haihe Biopharma Co., Ltd., Ignyta,Inc., Janssen Pharmaceutical K.K., KISSEI PHARMACEUTICAL CO., LTD., Kyowa Kirin Co., Ltd., Life Technologies Japan Ltd., Loxo Oncology, Inc., LSI Medience Corporation., MEDICAL& BIOLOGICAL LABORATORIES CO., LTD., Merck Biopharma Co., Ltd., Merus N.V., MSD K.K., Novartis Pharma K.K., Ono Pharmaceutical Co., Ltd., Pfizer Japan Inc., Pfizer R&D Japan G.K., Precision Medicine Asia Co., Ltd., RIKEN GENESIS CO., LTD., Sumitomo Pharma Co., Ltd., Spectrum Pharmaceuticals, Inc., Sysmex Corporation., Taiho Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd., Turning Point Therapeutics,Inc., Xcoo, Inc., honoraria for lecture from Amgen K.K., Amoy Diagnosties Co.,Ltd., AstraZeneca K.K., Bayer U.S., Boehringer Ingelheim Japan, Inc., Bristol-Myers Squibb K.K., Chugai Pharmaceutical Co., Ltd., DAIICHI SANKYO Co., Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., Guardant Health Inc., Janssen Pharmaceutical K.K., Merck Biopharma Co., Ltd., Nippon Kayaku Co.,Ltd., Novartis Pharma K.K., Ono Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Co., Ltd., RIKEN GENESIS CO., LTD., Taiho Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd., Thermo Fisher Scientific K.K., participation on a Data Safety Monitoring Board or Advisory Board from Amgen Inc., Amgen K.K., DAIICHI SANKYO Co., Ltd., Eli Lilly Japan K.K., Haihe Biopharma Co., Ltd., Janssen Pharmaceutical K.K., SYNEOS HEALTH CLINICAL K.K. Other authors declare no conflicts of interest.
Figures

Similar articles
-
Usefulness of endoscopic ultrasound with bronchoscope-guided fine-needle aspiration for next-generation sequencing in patients with non-small cell lung cancer: A comparison with other bronchoscopic techniques.Respir Investig. 2024 Sep;62(5):879-883. doi: 10.1016/j.resinv.2024.07.012. Epub 2024 Aug 2. Respir Investig. 2024. PMID: 39096541
-
Feasibility of Endobronchial Ultrasound-guided Transbronchial Needle Aspiration Cytology Specimens for Next Generation Sequencing in Non-small-cell Lung Cancer.Clin Lung Cancer. 2018 May;19(3):230-238.e2. doi: 10.1016/j.cllc.2017.11.010. Epub 2017 Dec 5. Clin Lung Cancer. 2018. PMID: 29277564
-
The Performance of an Extended Next Generation Sequencing Panel Using Endobronchial Ultrasound-Guided Fine Needle Aspiration Samples in Non-Squamous Non-Small Cell Lung Cancer: A Pragmatic Study.Clin Lung Cancer. 2023 Mar;24(2):e105-e112. doi: 10.1016/j.cllc.2022.11.010. Epub 2022 Dec 5. Clin Lung Cancer. 2023. PMID: 36599742 Free PMC article. Clinical Trial.
-
A systematic review and meta-analysis of the adequacy of endobronchial ultrasound transbronchial needle aspiration for next-generation sequencing in patients with non-small cell lung cancer.Lung Cancer. 2022 Apr;166:17-26. doi: 10.1016/j.lungcan.2022.01.018. Epub 2022 Jan 25. Lung Cancer. 2022. PMID: 35151114
-
Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA): an overview and update for the cytopathologist.Cancer Cytopathol. 2014 Aug;122(8):561-76. doi: 10.1002/cncy.21431. Epub 2014 Apr 23. Cancer Cytopathol. 2014. PMID: 24760496 Review.
Cited by
-
Suitability of Frozen Pleural Fluid Pellets for Next-Generation Sequencing-Based Driver Gene Testing in Non-Small Cell Lung Cancer.Thorac Cancer. 2025 Jun;16(11):e70107. doi: 10.1111/1759-7714.70107. Thorac Cancer. 2025. PMID: 40492645 Free PMC article.
References
-
- Lindeman NI, Cagle PT, Aisner DL, Arcila ME, Beasley MB, Bernicker EH, et al. Updated molecular testing guideline for the selection of lung cancer patients for treatment with targeted tyrosine kinase inhibitors: guideline from the college of American pathologists, the international association for the study of lung cancer, and the association for molecular pathology. J Thorac Oncol. 2018;13(3):321–46. doi: 10.1016/j.jtho.2017.12.001. - DOI - PubMed
-
- Kalemkerian GP, Narula N, Kennedy EB, Biermann WA, Donington J, Leighl NB, et al. Molecular testing guideline for the selection of patients with lung cancer for treatment with targeted tyrosine kinase inhibitors: American society of clinical oncology endorsement of the college of American pathologists/international association for the study of lung cancer/association for molecular pathology clinical practice guideline update. J Clin Oncol. 2018;36(9):911–9. doi: 10.1200/JCO.2017.76.7293. - DOI - PubMed
-
- Mok T, Camidge DR, Gadgeel SM, Rosell R, Dziadziuszko R, Kim DW, et al. Updated overall survival and final progression-free survival data for patients with treatment-naive advanced ALK-positive non-small-cell lung cancer in the ALEX study. Ann Oncol. 2020;31(8):1056–64. doi: 10.1016/j.annonc.2020.04.478. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

