Theta-burst rTMS in schizophrenia to ameliorate negative and cognitive symptoms: study protocol for a double-blind, sham-controlled, randomized clinical trial
- PMID: 38632647
- PMCID: PMC11025264
- DOI: 10.1186/s13063-024-08106-9
Theta-burst rTMS in schizophrenia to ameliorate negative and cognitive symptoms: study protocol for a double-blind, sham-controlled, randomized clinical trial
Abstract
Background: Treatment effects of conventional approaches with antipsychotics or psychosocial interventions are limited when it comes to reducing negative and cognitive symptoms in schizophrenia. While there is emerging clinical evidence that new, augmented protocols based on theta-burst stimulation can increase rTMS efficacy dramatically in depression, data on similar augmented therapies are limited in schizophrenia. The different patterns of network impairments in subjects may underlie that some but not all patients responded to given stimulation locations.
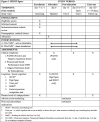
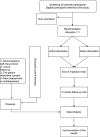
Methods: Therefore, we propose an augmented theta-burst stimulation protocol in schizophrenia by stimulating both locations connected to negative symptoms: (1) the left dorsolateral prefrontal cortex (DLPFC), and (2) the vermis of the cerebellum. Ninety subjects with schizophrenia presenting negative symptoms and aging between 18 and 55 years will be randomized to active and sham stimulation in a 1:1 ratio. The TBS parameters we adopted follow the standard TBS protocols, with 3-pulse 50-Hz bursts given every 200 ms (at 5 Hz) and an intensity of 100% active motor threshold. We plan to deliver 1800 stimuli to the left DLPFC and 1800 stimuli to the vermis daily in two 9.5-min blocks for 4 weeks. The primary endpoint is the change in negative symptom severity measured by the Positive and Negative Syndrome Scale (PANSS). Secondary efficacy endpoints are changes in cognitive flexibility, executive functioning, short-term memory, social cognition, and facial emotion recognition. The difference between study groups will be analyzed by a linear mixed model analysis with the difference relative to baseline in efficacy variables as the dependent variable and treatment group, visit, and treatment-by-visit interaction as independent variables. The safety outcome is the number of serious adverse events.
Discussion: This is a double-blind, sham-controlled, randomized medical device study to assess the efficacy and safety of an augmented theta-burst rTMS treatment in schizophrenia. We hypothesize that social cognition and negative symptoms of patients on active therapy will improve significantly compared to patients on sham treatment.
Trial registration: The study protocol is registered at "ClinicalTrials.gov" with the following ID: NCT05100888. All items from the World Health Organization Trial Registration Data Set are registered. Initial release: 10/19/2021.
Keywords: Cognitive symptoms; Negative symptoms; Schizophrenia; Theta-burst; Transcranial magnetic stimulation.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
References
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical