Response of human peripheral blood monocyte-derived macrophages (PBMM) to demineralized and decellularized bovine bone graft substitutes
- PMID: 38635511
- PMCID: PMC11025794
- DOI: 10.1371/journal.pone.0300331
Response of human peripheral blood monocyte-derived macrophages (PBMM) to demineralized and decellularized bovine bone graft substitutes
Abstract
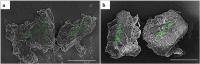
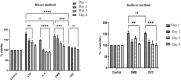
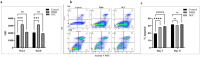
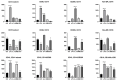
The performance of apparently biocompatible implanted bovine bone grafts may be compromised by unresolved chronic inflammation, and poor graft incorporation leading to implant failure. Monitoring the intensity and duration of the inflammatory response caused by implanted bone grafts is crucial. In this study, the ability of demineralized (DMB) and decellularized (DCC) bovine bone substitutes in initiating inflammatory responses to peripheral blood monocyte-derived macrophages (PBMMs) was investigated. The response of PBMMs to bone substitutes was evaluated by using both direct and indirect cell culture, reactive oxygen species (ROS) generation, apoptosis, immunophenotyping, and cytokine production. Analysis of DMB and DCC substitutes using scanning electron microscope (SEM) showed a roughened surface with a size ranging between 500 and 750 μm. PBMMs treated with DMB demonstrated cell aggregation and clumping mimicking lipopolysaccharide (LPS) treated PBMMs and a higher proliferation ability (166.93%) compared to control (100%) and DCC treatments (115.64%; p<0.001) at 24h. This was associated with a significantly increased production of intracellular ROS in PBMMs exposed to DMB substitutes than control (3158.5 vs 1715.5; p<0.001) and DCC treatment (2117.5). The bone substitute exposure also caused an increase in percentage apoptosis which was significantly (p<0.0001) higher in both DMB (27.85) and DCC (29.2) treatment than control (19.383). A significant increase in proinflammatory cytokine expression (TNF-α: 3.4 folds; p<0.05) was observed in DMB substitute-treated PBMMs compared to control. Notably, IL-1β mRNA was significantly higher in DMB (21.75 folds; p<0.0001) than control and DCC (5.01 folds). In contrast, DCC substitutes exhibited immunoregulatory effects on PBMMs, as indicated by the expression for CD86, CD206, and HLDR surface markers mimicking IL-4 treatments. In conclusion, DMB excites a higher immunological response compared to DCC suggesting decellularization process of tissues dampen down inflammatory reactions when exposed to PBMM.
Copyright: © 2024 Rani et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
No authors have competing interests.
Figures


Similar articles
-
A comparative study on immune responses to demineralized and decellularized bone substitute following intraperitoneal implantation in mouse model.PLoS One. 2025 May 16;20(5):e0323666. doi: 10.1371/journal.pone.0323666. eCollection 2025. PLoS One. 2025. PMID: 40378388 Free PMC article.
-
Evaluation of the osteogenic potential of demineralized and decellularized bovine bone granules following implantation in rat calvaria critical-size defect model.PLoS One. 2023 Dec 21;18(12):e0294291. doi: 10.1371/journal.pone.0294291. eCollection 2023. PLoS One. 2023. PMID: 38127838 Free PMC article.
-
Exploring the Biomaterial-Induced Secretome: Physical Bone Substitute Characteristics Influence the Cytokine Expression of Macrophages.Int J Mol Sci. 2021 Apr 24;22(9):4442. doi: 10.3390/ijms22094442. Int J Mol Sci. 2021. PMID: 33923149 Free PMC article.
-
Enoxaparin sodium bone cement plays an anti-inflammatory immunomodulatory role by inducing the polarization of M2 macrophages.J Orthop Surg Res. 2023 May 23;18(1):380. doi: 10.1186/s13018-023-03865-8. J Orthop Surg Res. 2023. PMID: 37221568 Free PMC article.
-
Origin of Monocytes/Macrophages Contributing to Chronic Inflammation in Chagas Disease: SIRT1 Inhibition of FAK-NFκB-Dependent Proliferation and Proinflammatory Activation of Macrophages.Cells. 2019 Dec 28;9(1):80. doi: 10.3390/cells9010080. Cells. 2019. PMID: 31905606 Free PMC article.
Cited by
-
Decoding biomaterial-associated molecular patterns (BAMPs): influential players in bone graft-related foreign body reactions.PeerJ. 2025 Apr 22;13:e19299. doi: 10.7717/peerj.19299. eCollection 2025. PeerJ. 2025. PMID: 40292103 Free PMC article. Review.
-
A comparative study on immune responses to demineralized and decellularized bone substitute following intraperitoneal implantation in mouse model.PLoS One. 2025 May 16;20(5):e0323666. doi: 10.1371/journal.pone.0323666. eCollection 2025. PLoS One. 2025. PMID: 40378388 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

