Photoresponse of new azo pyridine functionalized poly(2-hydroxyethyl methacrylate-co-methyl methacrylate)
- PMID: 38643277
- PMCID: PMC11032328
- DOI: 10.1038/s41598-024-59704-1
Photoresponse of new azo pyridine functionalized poly(2-hydroxyethyl methacrylate-co-methyl methacrylate)
Abstract
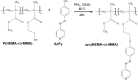
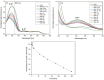
A new azo polymer containing photoisomerizable azo pyridine functionalities was synthesized via Mitsunobu reaction of 4-(4-hydroxyphenylazo)pyridine with poly(2-hydroxyethyl methacrylate-co-methyl methacrylate) (p(HEMA-co-MMA)) for creating new photochromic materials. The resulting polymer with azo pyridine side groups was characterized for structural, thermal, and optical properties. UV-vis, 1H NMR and IR spectroscopies confirmed that all hydroxyl groups in p(HEMA-co-MMA) were substituted with azo dye. The obtained azo copolymer exhibited high thermal stability (around 240 °C) and a glass transition temperature (113 °C), promising for applications. The trans-to-cis isomerization upon UV irradiation and the thermal back reaction of the azo chromophore in the copolymer in the solid state was studied. A photostationary state with 50% content of cis-isomers upon 6 min of UV irradiation was reached, and during 48 h dark relaxation at ambient temperature, all cis-isomers converted to the trans form. Additionally, the possibility of efficient photogeneration of surface relief gratings with high amplitude of azo copolymer surface modulation was demonstrated.
© 2024. The Author(s).
Conflict of interest statement
The authors declare the following financial interests relationships which may be considered as potential competing interests: National Science Centre, Poland, grant number UMO-2019/35/D/ST5/00533.
Figures




Similar articles
-
Peptide surface modification of P(HEMA-co-MMA)-b-PIB-b-P(HEMA-co-MMA) block copolymers.Langmuir. 2009 Jun 2;25(11):6319-27. doi: 10.1021/la9000768. Langmuir. 2009. PMID: 19334689
-
Stimuli-Chromic Oxazolidine Latex Nanoparticles for Dual-Responsive pH-Sensors and Rewritable Halochromic Papers: A Physicochemical Study on Colorimetric and Fluorimetric Signals.ACS Appl Mater Interfaces. 2025 Jan 29;17(4):6987-7006. doi: 10.1021/acsami.4c22458. Epub 2025 Jan 20. ACS Appl Mater Interfaces. 2025. PMID: 39832795
-
Relationship between water structure and properties of poly(methyl methacrylate-b-2-hydroxyethyl methacrylate) by solid-state NMR.J Biomater Sci Polym Ed. 2017 Jul-Aug;28(10-12):1199-1214. doi: 10.1080/09205063.2017.1310647. Epub 2017 Apr 3. J Biomater Sci Polym Ed. 2017. PMID: 28325108
-
Optical properties of some new azo photoisomerizable bismaleimide derivatives.Int J Mol Sci. 2011;12(9):6176-93. doi: 10.3390/ijms12096176. Epub 2011 Sep 21. Int J Mol Sci. 2011. PMID: 22016652 Free PMC article.
-
Thermoresponsive Smart Copolymer Coatings Based on P(NIPAM-co-HEMA) and P(OEGMA-co-HEMA) Brushes for Regenerative Medicine.ACS Biomater Sci Eng. 2023 Nov 13;9(11):6256-6272. doi: 10.1021/acsbiomaterials.3c00917. Epub 2023 Oct 24. ACS Biomater Sci Eng. 2023. PMID: 37874897 Free PMC article.
Cited by
-
Recent Advances in the Mitsunobu and Related Reactions: A Review from 2010 to 2024.Top Curr Chem (Cham). 2025 Mar 18;383(2):15. doi: 10.1007/s41061-025-00501-3. Top Curr Chem (Cham). 2025. PMID: 40097732 Review.
References
-
- Węgłowski R, Piecek W, Kozanecka-Szmigiel A, Konieczkowska J, Schab-Balcerzak E. Poly(esterimide) bearing azobenzene units as photoaligning layer for liquid crystals. Opt. Mater. 2015;49:224. doi: 10.1016/j.optmat.2015.09.020. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

