Interleukin-4 protects retinal ganglion cells and promotes axon regeneration
- PMID: 38650003
- PMCID: PMC11034112
- DOI: 10.1186/s12964-024-01604-y
Interleukin-4 protects retinal ganglion cells and promotes axon regeneration
Abstract
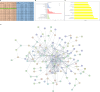
Background: The preservation of retinal ganglion cells (RGCs) and the facilitation of axon regeneration are crucial considerations in the management of various vision-threatening disorders. Therefore, we investigate the efficacy of interleukin-4 (IL-4), a potential therapeutic agent, in promoting neuroprotection and axon regeneration of retinal ganglion cells (RGCs) as identified through whole transcriptome sequencing in an in vitro axon growth model.
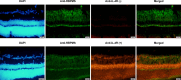
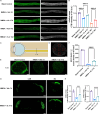
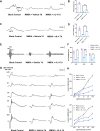
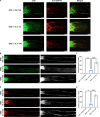
Methods: A low concentration of staurosporine (STS) was employed to induce in vitro axon growth. Whole transcriptome sequencing was utilized to identify key target factors involved in the molecular mechanism underlying axon growth. The efficacy of recombinant IL-4 protein on promoting RGC axon growth was validated through in vitro experiments. The protective effect of recombinant IL-4 protein on somas of RGCs was assessed using RBPMS-specific immunofluorescent staining in mouse models with optic nerve crush (ONC) and N-methyl-D-aspartic acid (NMDA) injury. The protective effect on RGC axons was evaluated by anterograde labeling of cholera toxin subunit B (CTB), while the promotion of RGC axon regeneration was assessed through both anterograde labeling of CTB and immunofluorescent staining for growth associated protein-43 (GAP43).
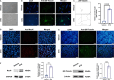
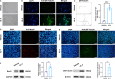
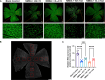
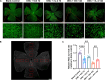
Results: Whole-transcriptome sequencing of staurosporine-treated 661 W cells revealed a significant upregulation in intracellular IL-4 transcription levels during the process of axon regeneration. In vitro experiments demonstrated that recombinant IL-4 protein effectively stimulated axon outgrowth. Subsequent immunostaining with RBPMS revealed a significantly higher survival rate of RGCs in the rIL-4 group compared to the vehicle group in both NMDA and ONC injury models. Axonal tracing with CTB confirmed that recombinant IL-4 protein preserved long-distance projection of RGC axons, and there was a notably higher number of surviving axons in the rIL-4 group compared to the vehicle group following NMDA-induced injury. Moreover, intravitreal delivery of recombinant IL-4 protein substantially facilitated RGC axon regeneration after ONC injury.
Conclusion: The recombinant IL-4 protein exhibits the potential to enhance the survival rate of RGCs, protect RGC axons against NMDA-induced injury, and facilitate axon regeneration following ONC. This study provides an experimental foundation for further investigation and development of therapeutic agents aimed at protecting the optic nerve and promoting axon regeneration.
Keywords: Axon regeneration; Nerve excitotoxicity; Optic nerve crush; Retinal ganglion cells.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Tumor Necrosis Factor Receptor Superfamily Member 12A Enhances Retinal Ganglion Cell Survival and Promotes Axon Regeneration.FASEB J. 2025 Aug 15;39(15):e70895. doi: 10.1096/fj.202500988RR. FASEB J. 2025. PMID: 40747811
-
Co-delivery of neurotrophic factors and a zinc chelator substantially increases retinal ganglion cell survival and axon protection in the optic nerve crush model.Acta Biomater. 2025 Jul 1;201:297-308. doi: 10.1016/j.actbio.2025.06.007. Epub 2025 Jun 5. Acta Biomater. 2025. PMID: 40482982 Free PMC article.
-
Tauroursodeoxycholic Acid Protects Retinal Ganglion Cells and Reduces Inflammation in Mice Following Optic Nerve Crush.Pharmaceuticals (Basel). 2025 Apr 14;18(4):569. doi: 10.3390/ph18040569. Pharmaceuticals (Basel). 2025. PMID: 40284004 Free PMC article.
-
Retinal ganglion cell repopulation for vision restoration in optic neuropathy: a roadmap from the RReSTORe Consortium.Mol Neurodegener. 2023 Sep 21;18(1):64. doi: 10.1186/s13024-023-00655-y. Mol Neurodegener. 2023. PMID: 37735444 Free PMC article. Review.
-
Neuroprotection for treatment of glaucoma in adults.Cochrane Database Syst Rev. 2013 Feb 28;2(2):CD006539. doi: 10.1002/14651858.CD006539.pub3. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2017 Jan 25;1:CD006539. doi: 10.1002/14651858.CD006539.pub4. PMID: 23450569 Free PMC article. Updated.
Cited by
-
Protocol for the purification and culture of primary retinal ganglion cells and development of common pathological models.J Mol Histol. 2025 Aug 8;56(4):260. doi: 10.1007/s10735-025-10536-x. J Mol Histol. 2025. PMID: 40779178
-
Deletion of Slc1a4 Suppresses Single Mauthner Cell Axon Regeneration In Vivo through Growth-Associated Protein 43.Int J Mol Sci. 2024 Oct 11;25(20):10950. doi: 10.3390/ijms252010950. Int J Mol Sci. 2024. PMID: 39456733 Free PMC article.
-
AAV2.7m8 transduction of stage 2 human retinal organoids induces highly variable responses in innate and inflammatory gene expression and cytokine secretion.Exp Eye Res. 2025 Sep;258:110478. doi: 10.1016/j.exer.2025.110478. Epub 2025 Jun 6. Exp Eye Res. 2025. PMID: 40484363
References
Publication types
MeSH terms
Substances
Grants and funding
- No. 82171053, 81570864/National Natural Science Foundation of China
- No. 82171053, 81570864/National Natural Science Foundation of China
- No. 82171053, 81570864/National Natural Science Foundation of China
- No. 82171053, 81570864/National Natural Science Foundation of China
- No. 82171053, 81570864/National Natural Science Foundation of China
LinkOut - more resources
Full Text Sources

