Cannabidiol potentiates hyperpolarization-activated cyclic nucleotide-gated (HCN4) channels
- PMID: 38652080
- PMCID: PMC11040500
- DOI: 10.1085/jgp.202313505
Cannabidiol potentiates hyperpolarization-activated cyclic nucleotide-gated (HCN4) channels
Abstract
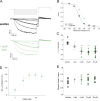
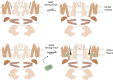
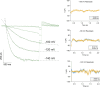
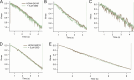
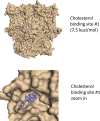
Cannabidiol (CBD), the main non-psychotropic phytocannabinoid produced by the Cannabis sativa plant, blocks a variety of cardiac ion channels. We aimed to identify whether CBD regulated the cardiac pacemaker channel or the hyperpolarization-activated cyclic nucleotide-gated channel (HCN4). HCN4 channels are important for the generation of the action potential in the sinoatrial node of the heart and increased heart rate in response to β-adrenergic stimulation. HCN4 channels were expressed in HEK 293T cells, and the effect of CBD application was examined using a whole-cell patch clamp. We found that CBD depolarized the V1/2 of activation in holo-HCN4 channels, with an EC50 of 1.6 µM, without changing the current density. CBD also sped activation kinetics by approximately threefold. CBD potentiation of HCN4 channels occurred via binding to the closed state of the channel. We found that CBD's mechanism of action was distinct from cAMP, as CBD also potentiated apo-HCN4 channels. The addition of an exogenous PIP2 analog did not alter the ability of CBD to potentiate HCN4 channels, suggesting that CBD also acts using a unique mechanism from the known HCN4 potentiator PIP2. Lastly, to gain insight into CBD's mechanism of action, computational modeling and targeted mutagenesis were used to predict that CBD binds to a lipid-binding pocket at the C-terminus of the voltage sensor. CBD represents the first FDA-approved drug to potentiate HCN4 channels, and our findings suggest a novel starting point for drug development targeting HCN4 channels.
© 2024 Page and Ruben.
Conflict of interest statement
Disclosures: D.A. Page reported grants from Akseera Pharma Corp during the conduct of the study. P.C. Ruben reported grants from MITACS/Akseera Pharma, Inc. during the conduct of the study.
Figures





Similar articles
-
Long-QT syndrome-associated caveolin-3 mutations differentially regulate the hyperpolarization-activated cyclic nucleotide gated channel 4.Physiol Int. 2017 Jun 1;104(2):130-138. doi: 10.1556/2060.104.2017.2.6. Epub 2017 Jun 26. Physiol Int. 2017. PMID: 28648120
-
Phosphorylation and modulation of hyperpolarization-activated HCN4 channels by protein kinase A in the mouse sinoatrial node.J Gen Physiol. 2010 Sep;136(3):247-58. doi: 10.1085/jgp.201010488. Epub 2010 Aug 16. J Gen Physiol. 2010. PMID: 20713547 Free PMC article.
-
A mechanism for the auto-inhibition of hyperpolarization-activated cyclic nucleotide-gated (HCN) channel opening and its relief by cAMP.J Biol Chem. 2014 Aug 8;289(32):22205-20. doi: 10.1074/jbc.M114.572164. Epub 2014 May 30. J Biol Chem. 2014. PMID: 24878962 Free PMC article.
-
Pacemaker activity of the human sinoatrial node: effects of HCN4 mutations on the hyperpolarization-activated current.Europace. 2014 Mar;16(3):384-95. doi: 10.1093/europace/eut348. Europace. 2014. PMID: 24569893 Review.
-
The structure of the apo cAMP-binding domain of HCN4 - a stepping stone toward understanding the cAMP-dependent modulation of the hyperpolarization-activated cyclic-nucleotide-gated ion channels.FEBS J. 2018 Jun;285(12):2182-2192. doi: 10.1111/febs.14408. Epub 2018 Mar 14. FEBS J. 2018. PMID: 29444387 Review.
Cited by
-
Impact of Long-Term Cannabidiol (CBD) Treatment on Mouse Kidney Transcriptome.Genes (Basel). 2024 Dec 21;15(12):1640. doi: 10.3390/genes15121640. Genes (Basel). 2024. PMID: 39766907 Free PMC article.
-
Endocannabinoid regulation of inward rectifier potassium (Kir) channels.Front Pharmacol. 2024 Aug 26;15:1439767. doi: 10.3389/fphar.2024.1439767. eCollection 2024. Front Pharmacol. 2024. PMID: 39253376 Free PMC article.
-
Pharmacology of Non-Psychoactive Phytocannabinoids and Their Potential for Treatment of Cardiometabolic Disease.Handb Exp Pharmacol. 2025;287:61-93. doi: 10.1007/164_2024_731. Handb Exp Pharmacol. 2025. PMID: 39235486 Review.
References
-
- Akimoto, M., Zhang Z., Boulton S., Selvaratnam R., VanSchouwen B., Gloyd M., Accili E.A., Lange O.F., and Melacini G.. 2014. A mechanism for the auto-inhibition of hyperpolarization-activated cyclic nucleotide-gated (HCN) channel opening and its relief by cAMP. J. Biol. Chem. 289:22205–22220. 10.1074/jbc.M114.572164 - DOI - PMC - PubMed
-
- Baruscotti, M., Bucchi A., Viscomi C., Mandelli G., Consalez G., Gnecchi-Rusconi T., Montano N., Casali K.R., Micheloni S., Barbuti A., and DiFrancesco D.. 2011. Deep bradycardia and heart block caused by inducible cardiac-specific knockout of the pacemaker channel gene Hcn4. Proc. Natl. Acad. Sci. USA. 108:1705–1710. 10.1073/pnas.1010122108 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

